Crystals are what happens when matter gets organized. Atoms, ions, or molecules line up in repeating patterns, building solid structures that can be seen with the naked eye or only under a microscope. This invisible order is what gives crystals their characteristic shapes, colors, and properties, whether they form deep underground, in a metal pot of molten bismuth, or in a jar of salty water on a kitchen counter.
What a crystal really is
A crystal is a solid in which the building blocks—atoms, ions, or molecules—repeat in an orderly three‑dimensional pattern called a crystal lattice. This regularity distinguishes crystalline solids from amorphous ones like glass, where the particles are more randomly arranged.
Because of this internal order, crystals often grow with flat faces and characteristic angles that reflect their underlying lattice. Salt, for example, tends to form cubes because sodium and chloride ions stack in a cubic arrangement, while quartz grows hexagonal prisms based on its silicon–oxygen framework.
How crystals form from solutions
One of the most accessible ways to form crystals—great for kids and labs alike—is from a solution. When a solute such as salt is dissolved in water, there is a limit to how much can dissolve at a given temperature; that limit is called the solubility.
Crystallization usually follows three stages.
-
First, a saturated or supersaturated solution forms, often by dissolving more material at high temperature and then cooling it.
-
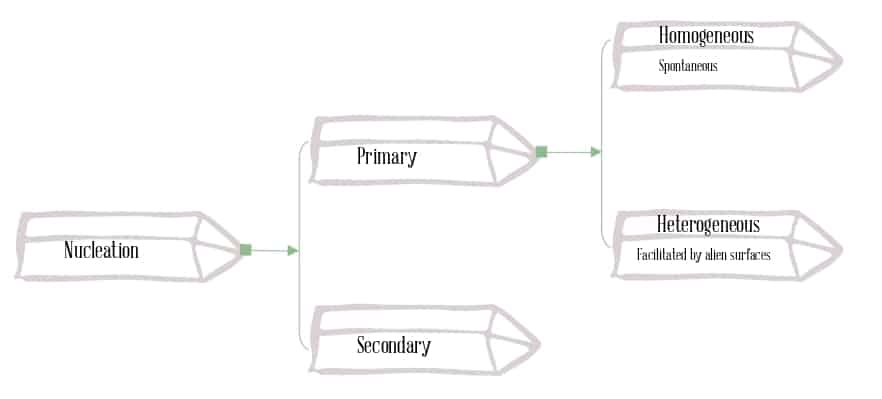
Second, nucleation occurs when a few ions or molecules cluster together in a stable way, creating a tiny “seed” crystal.
-
Third, crystal growth proceeds as more particles attach to that seed, building visible structures that reflect the chemistry and conditions of the system.
A supersaturated solution is a key teaching tool: it contains more dissolved material than would normally be stable at that temperature, so it is “primed” to crystallize as soon as nucleation is triggered by a disturbance, seed crystal, or surface. This is the same basic chemistry behind growing salt ornaments, sodium acetate “hot ice,” and many classic classroom crystal projects .
Crystals from melts: bismuth and beyond
Crystals can also form directly from a melt, which is the basis of your bismuth crystal activity. In this case, the atoms are in a liquid metal rather than a water solution, but the physics is similar: as the liquid cools, atoms lose energy and begin to lock into an orderly lattice.
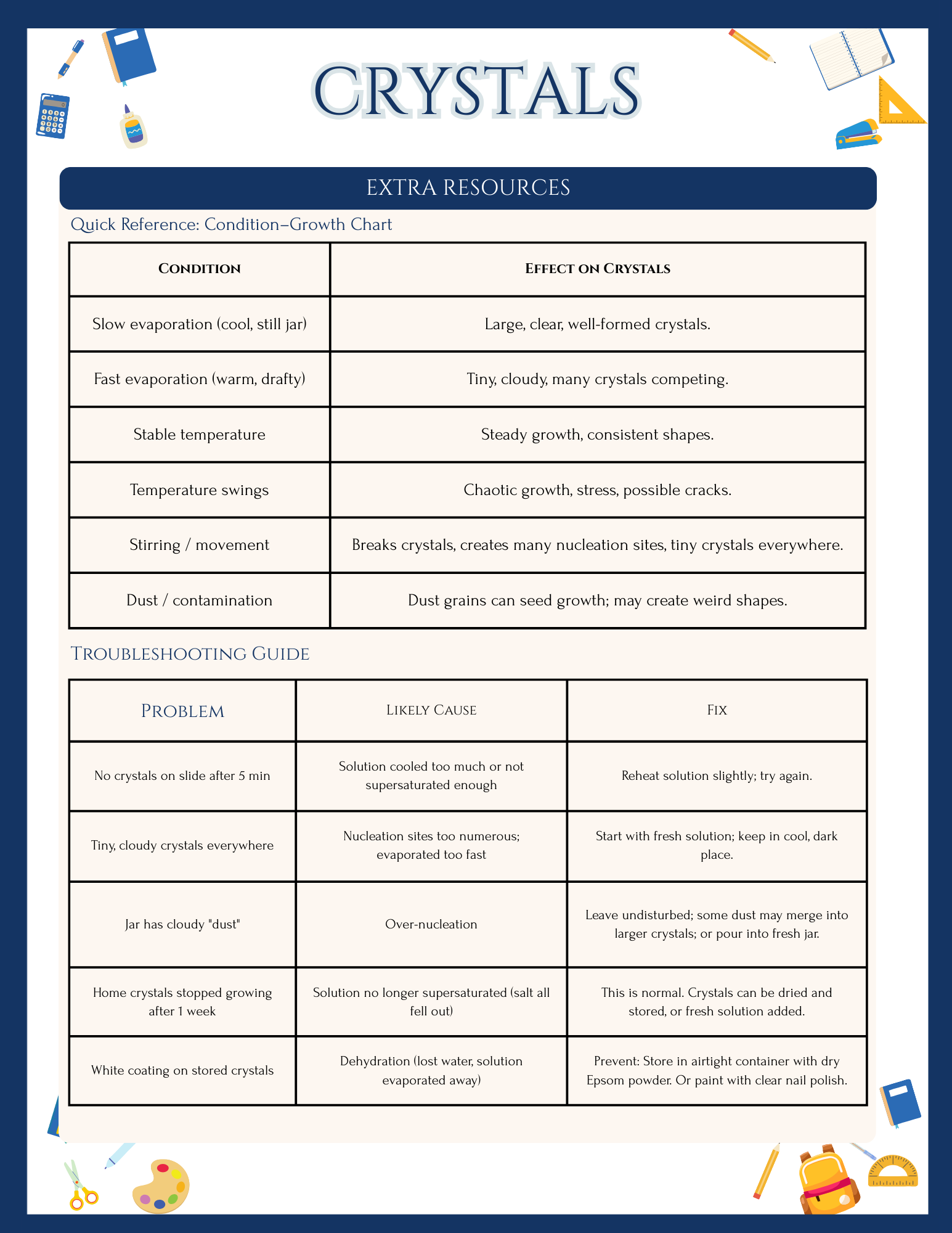
Bismuth has a relatively low melting point for a metal and crystallizes in stepped, geometric hopper crystals with iridescent oxide layers that make it visually striking for students. Cooling rate matters: slower cooling generally allows fewer nuclei to form and gives the existing crystals more time to grow, leading to larger, more dramatic structures; faster cooling yields many small crystals. These same principles are central to industrial metallurgy, where controlling cooling conditions tunes grain size and therefore strength, brittleness, and other mechanical properties in metals and alloys.
Why shape and structure matter
The internal structure of a crystal controls its external shape and many of its physical properties. The orientation and spacing of lattice planes determine cleavage directions—the planes along which minerals tend to break—which is a core concept in geology and materials science.
Crystalline order also affects hardness, optical behavior, and electrical properties. For example, quartz and other piezoelectric crystals generate a voltage when mechanically stressed, enabling timing in watches and sensors. There can even be multiple crystal forms (polymorphs) of the same substance—like different arrangements of the same chemical formula—each with distinct stability and behavior, which is crucial in areas such as pharmaceuticals .
Everyday crystals and industrial workhorses
Crystals quietly support modern life. Some examples that connect well with kids and parents:
-
Food and household: Table salt, sugar, and many food additives are crystalline materials, and their crystal size and shape influence texture and how they dissolve.
-
Electronics: Semiconductor devices rely on ultrapure single crystals of silicon and related materials; defects or disorder in the crystal lattice can disrupt how electrons move and degrade device performance.
-
Construction and infrastructure: Metals in bridges, cars, and tools are crystalline; controlled crystal size and orientation in steel and other alloys are what give them specific strength and toughness.
-
Medicine and chemistry: Many drugs are crystallized to obtain a defined form with predictable solubility and stability, and x‑ray diffraction of crystals has been a central method for determining molecular structures.
Even the “crystalline art to tasty salts” style activities are rooted in the same underlying science: a controlled environment that nudges molecules or ions to organize into repeatable patterns, whether for aesthetics, flavor, or function.

Why crystals make great teaching tools
Crystals are ideal for science education because they bridge the gap between the invisible atomic world and what students can actually see and handle. A simple crystal-growing setup demonstrates key concepts in chemistry and physics:
-
Particle behavior and solubility: Heating and cooling show how temperature affects solubility and particle motion.
-
Phase changes and energy: Melting and solidifying bismuth, or triggering crystallization from a supersaturated solution, highlight how energy is absorbed or released as matter changes state.
-
Structure–property relationships: Viewing crystals under a microscope reinforces that macro‑scale shape and properties come from micro‑scale order.
This kind of hands-on, visual activity also supports inquiry skills: students can ask, test, and observe how changing one variable—such as cooling rate, concentration, or type of solute—affects the crystals they grow.
Connecting to Insanitek labs and what’s next
The earlier Insanitek posts on how crystals form in supersaturated solutions, making bismuth crystals, and creating crystalline art and salts give readers multiple entry points into the same core science: order emerging from chaos through carefully tuned conditions. Together, they show that crystal chemistry is:
-
Deeply rooted in real thermodynamics and kinetics
-
Accessible enough for kitchen‑table experiments
-
Powerful enough to underpin electronics, materials, and medicine
Below is the lesson plan and activity to the lab we did, hosted by Arrows Learning Academy in Indianapolis, where students grew crystals and then study them under a microscope, tying visual observations back to solubility, nucleation, growth, and structure–property relationships. The full lesson plan and activity guide walk through these steps explicitly so families and educators can replicate (or remix) the experience and keep using crystals as a gateway into serious, curiosity‑driven science — even at birthday parties.
Crystal Systems & Epsom Salt Growing Lab
$ 7.00 Price excludes tax and S&H
A ready-to-print crystal science lab for ages 7–15 that combines a full lesson plan, student worksheets, vocab cards, and an at‑home Epsom salt crystal‑growing experiment. Learners explore crystal systems, observe real crystals under the microscope, and grow their own crystals over 1–2 weeks using clear, parent‑ and teacher‑friendly guidance
Use it as a stand‑alone crystal lab or a 1–2 week mini‑unit on crystals and the structure of matter.
Description
Turn your kitchen table, classroom, or church basement into a crystal lab with this ready‑to‑run STEM experience for ages 7–15. This digital kit bundles a full lesson plan, hands‑on activities, printable worksheets, and vocabulary cards so learners can explore crystal shapes, grow their own crystals, and talk about them using real science language.
Product Description
The Crystal Systems & Epsom Salt Lab Pack gives you everything needed to run a complete crystals lesson without starting from scratch. Students learn how atoms arrange into different crystal systems, observe Epsom salt crystals forming under a microscope, and then take home a crystal seed jar to grow over 1–2 weeks. Materials are written for mixed ages (7–15) and work in after‑school clubs, homeschool co‑ops, church programs, and classrooms.
What’s Included
-
Full Lesson Plan (Teacher/Parent Guide)
-
Clear learning targets and NGSS connections for upper elementary and middle school.
-
Suggested 2‑hour flow with two built‑in breaks, including when to teach shapes, when to use microscopes, and when to set up take‑home jars.
-
Materials list, safety notes for working with glass and warm solutions, and core talking points linking crystal shape to atomic structure.
-
-
Student Worksheets & Activities
-
Crystal systems reference page (cubic, tetragonal, orthorhombic, hexagonal/trigonal, monoclinic, triclinic) with kid‑friendly descriptions.
-
“Match the Crystal Shape” pages where students examine real rocks/minerals and record which system they see.
-
Microscope slide page for sketching Epsom salt crystals as they appear while a drop dries, plus prompts about what makes something a crystal.
-
At‑home crystal growth log so students can track daily changes in their seed crystal jar for 1–2 weeks.
-
-
At‑Home Epsom Salt Crystal Protocol (Family Handout)
-
Clear, step‑by‑step care instructions for the take‑home jar, using common household locations (pantry, closet, bookshelf).
-
Guidance on what to expect during the first days and weeks, when to be patient, and how to preserve finished crystals.
-
Troubleshooting tips for tiny, cloudy, or stalled crystals.
-
-
Vocabulary Cards (Printable)
-
Ready‑to‑cut cards featuring key terms such as crystal, crystal system, cubic, hexagonal, orthorhombic, supersaturated solution, nucleation, acicular, and more.
-
Each card includes a simple definition and an in‑context example sentence that connects directly to the lab (“Epsom salt forms needle‑like orthorhombic crystals in this experiment.”).
-
Flexible for games (matching, sorting by crystal system, exit tickets, or word walls) in both classroom and club settings.
-
-
Assessment & Reflection Tools
-
Low‑prep options: observation checklists, quick exit questions, and simple drawing checks you can do in minutes.
-
Deeper options: short quizzes on crystal systems, sketch rubrics, and optional lab report prompts for older students who want more challenge.
-
Key Features
-
Plug‑and‑play: Just add Epsom salt, water, and basic lab supplies—the concepts, prompts, and printables are ready.
-
Mixed‑age friendly: Built‑in supports for younger learners plus extension questions for older students, all in one coherent package.
-
Vocabulary‑rich: Vocab cards, in‑context usage, and notebook prompts help students speak and write like young mineralogists.
-
Printer‑friendly: All pages are designed for black‑and‑white printing and easy photocopying for clubs, classrooms, and co‑ops.
Use this pack as a stand‑alone “wow” crystal day or as the backbone of a short unit on crystals, minerals, and the structure of matter.
Only logged in customers who have purchased this product may leave a review.





























Reviews
There are no reviews yet.