Oktoberfest, a fun-fest held throughout the world, started as celebration to honour the Crown Prince, Ludwig, and Princess Therese of Saxony-Hildburghausen on the 12th of October in 1810. The marriage celebration was held out in the Weis’n fields that are just outside of the palace. There were days of merriment, but the part that mattered the most were the horse races that signalled the close to the event. When the royal couple decided that the horse races and other festivities were to be repeated the next year, Oktoberfest was officially born.
Oktoberfest is one of the world’s largest festivals that is celebrated on a small-scale where ever there is a love of beer and German culture. In Germany, however, Oktoberfest, held from late September to the first weekend in October, is likely to see some 6 million people converge to celebrate the annual celebration full of laughter, food, drinking, games, and of course, horse races. The biggest part of the celebration now, though, is beer.
The rise of the brew
In any major city you’ll be able to find a microbrewery or more, each making their own artisan craft beer. It wasn’t always this way, though. As recently as the 1980s there were not a lot of interest in a good brew, so everything was mass-produced. Not a bad thing, but as everything works, we desire change. Slowly, people started to craft their own, sharing it with friends, and having a good time. At the turn of the century, the UK had only 500 breweries, but by 2012, they had added another 160 to their list (and still counting). In 2013 the US held 2,768 craft breweries on top of the normal non-craft breweries.
This, of course, brought back the home brewing revolution, and the rise of supply shops like Great Fermentations in Indianapolis, Indiana. Great Fermentations not only sells a plethora of supplies, but also holds classes on how to make great brew and wine at home. According to the Brewers Association, craft brewing increased 18% from 2012 to 2013, and 2014 is sure to see another increase.

With home brewing on the rise, Insanitekians, with the help of Tom McNay and Wes Martin of Great Fermentations, have decided to celebrate by exploring the science and art of brewing a great beer.
The science behind the art
From the water to the flavourings added, there are many variables to the science of brewing. Every part of the process can change the characteristics and flavour if the beer, which is what makes it an art. The grains make a base difference, but the minerals in the water add an elemental taste to the beer that can speak of location, while a distilled water can highlight added flavours.
Everything from the type of hops to the yeast, the water and the grains can change the flavour of the brew making it distinctly yours. That’s what makes them an art and a craft.
Behind the art is a science. Let’s explore.
The difference a little H20 makes

The first thing Tom McNay, sales associate of Great Fermentations and home brewer, told us was sterilisation is the key. You will use 3 times as much water in cleaning as you do in the process itself. This means that if you sterilise your equipment thoroughly, you’ll have only the favours you decide should be in there.
This, of course, starts with the water itself. The minerals in the water can be either natural or added. Either way you get them, they are called “brewing salts”, and make up the water profile of the beer, which can control the taste to a great degree. A higher gypsum content in the water increases the hop taste, while the calcium carbonate increase the malty flavour.
In other words, you can’t make a good brew with a foul-tasting water. You must treat it with a variety of treatments that can change the way a taste. One way to accomplish this is to start with your water report. According to How to Brew by John J. Palmer, you should be able to get your city’s water report from the Public Works department at your local City Hall (at least in the States) free of charge.
Once you have your water report, you can analyse your water and see what you need to change it. The mash pH of beer should be around 5.1 to 5.5 when measured at mash temperature (5.4 to 5.8 at room temperature), higher for roasted speciality malts. In the end the ion makeup of the water and the overall pH will determine how much of an effect each malt addition has. The goal in the art of home brewing, as we know, is to balance these flavours of the water and malt.
Water is the foundation of your brew, so understanding the chemistry make-up of your particular area, how to modify it, and how to work with the malts to enhance the flavours of your brew is a must.
Water isn’t everything, though.
Malting the grains
The first step of brewing is soaking the grains in warm water, which causes them to sprout slightly (malting). This changes the enzymes and sugars slightly and gives the grains the malted flavour, but more importantly, releases some of the starches used for fermenting. After the grains have sprouted, they are crushed and ground to extract the full flavour of the malt as well as the associated enzymes and sugars that will ferment more easily.
Sounds easy enough, doesn’t it? Well, it is easy, but the science behind it is a little more complicated than that. When the grain is waiting to sprout, the nutrients (starch) are enclosed in the husks of the barley grain. When the grain starts to sprout, this impregnable shell is breached, and the yeast can get to the starches within. Biologically speaking, this is the stage when the plant would be converting all it’s reserves in the seed for its own use to grow into a plant. The maltster gauges the germination carefully so that they achieve the right balance between the seeds resources being converted by the plant’s shoot and the amount it actually consumes for growth.
While this process begins to break down the seed, it is a very small amount. But why make the yeast struggle for so little starch? Grinding the grains after malting them releases the rest of the starches so the yeast can truly have a feast. Once the grains are malted, then ground, it’s time to focus on the mash.
Mash makin’
Mashing is a huge step in the beer making process that really, just about everything happens. It is here that starches are converted to fermentable sugars, primarily maltose. It’s the phase of brewing where your flavours begin to form from the water profile and grain types. water profile becomes really important, because as you mix the grains with hot water, it infuses the beer with a different flavour based on your water profile (among other factors). This step, essentially, is where the beer is made.
The enzymes, amylases, that are released during the malting process broke down the protein of the cell walls, releasing the starches to the water and hungry yeast, but it does something more during this phase of brewing. A malt has a diastatic power, or a measure of how much starch converting power a barley has, and it starts at the mashing stage. Diastatic power is measured in degrees Lintner (often shortened to °L), or in Europe as Windisch-Kolbach units (°WK) is often used. Without a high enough diastatic power , you won’t have as many sugars as you need to make a good brew. So how can you tell?
As it turns out, grains are often published with their Lintner or Windisch-Kolbach value in the malt supplier books. You can easily convert between the two using Lintner=(WK+16)/3.5 or going the other way as WK=3.5*Lintner 16. A malt needs a diastatic power of approximately 35 °L to be considered self converting. When a grain doesn’t have that, it must be supplemented somehow with other sugars to ferment fully, usually by adding rice, corn, or other refined sugars. On the other hand, malts with no diastatic power are used to flavour the brews.
And, this brings us to the wonders of yeast.
Yeasties!
Yeast is a single cell fungus, of which there are practically amazing. While I’d love to describe it further, no one can possibly describe how yeast works better than Alton Brown of Good Eats. (For a better quality video, you can buy and download it from Amazon Prime Good Eats Season 8. This was from Dr. Strangeloaf.)
Did you notice that he said that different yeasts from different locations have a different flavour? As in bread making and in beer making, yeast can impart a flavour into the mix that can make it uniquely yours. One thing to note about flavours is that as yeast ruptures, it can release several “off” flavours, such as sulphur and rubber. If you have a large mass of yeast on the bottom of a fermenter, you have a much higher potential for the yeast releasing these unpleasant and uncontrollable flavours.
This natural process is called autolysis, and it is often encountered by making a lager. A lager wins the yeast battle because the brewer removes the dead yeast and allows the brew to set for a second fermentation in a long, cold storage. This cool storage (45 – 55 ºF/ 7 – 13 °C)
- inhibits the formation of fruity esters by the yeast
- provides a longer conditioning period for the yeast to finish residual sugars and metabolise off flavours and aromas
- gives time for the haze-producing proteins and tannins to settle out, resulting in a clear beer
With lagering, the key is to maintain a balance so the autolysis doesn’t have the chance to produce off flavours and aromas that can ruin the brew.
Hops
We’ve covered grains, sugars, and yeasts. We’ve even noted the small, but crucial elements of minerals and proteins. But there is one more huge element we need to go through. Hops.
Hops are more than just flavours that one adds to the beer. Hops were likely introduced into beer for their preservative properties. The chemical in hops that is responsible for preserving and stabilising the beer is lupulin, which has antibacterial properties that make all this possible. Hops are so diverse in their purpose and flavour, that many hybrids are being made today. But what about them makes them so useful to beer, and how, exactly, do they work?
The hop cone contains 3 different resins. Two “soft” resins are alpha and beta, while gamma resins are “hard”. Soft resins add the aromatic agents, and not much is known about the hard resin, but it seems to lend to the bitter tastes that hops gives to beer. Of these resins, the alpha resin is intensely bitter, and becomes soluble as alpha acids during the boiling of wort. During a long boil, these alpha acids are isomerised and become even more bitter. — hence the term “bittering” hops.
Besides adding a bitter-sweet finish to the final brew, iso-alpha-acids also combine with tannins as there are unwanted proteins within the mix. When the proteins come into contact with the tannins, they combine. The combined molecules are then heavy enough to fall out of the solution as a precipitate, and remove proteins that can cloud the beer.
The beta resins, or essential oils within, are far less stable and evaporate quickly within the process. However, these resins are responsible for the hoppy flavour of beer when added later in the hopping schedule. Capturing these oils that give the beer a depth of character and aroma can be difficult for the initiated. Dry hopping is, perhaps, the most common and easiest way.
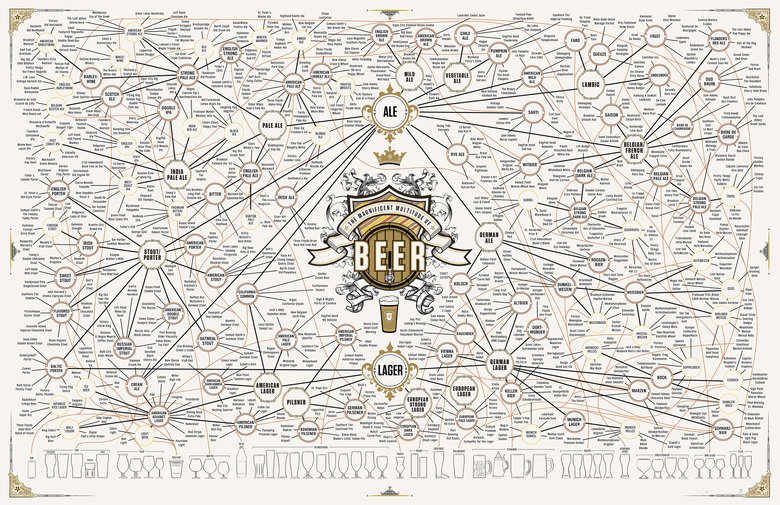
This year is the 181st Oktoberfest. How will you celebrate?
Oktoberfest is a great time of year to celebrate the harvest, indulge in a human past-time of making and/or drinking beer. We Insanitekians plan to celebrate by going to a pub and raising a glass. How will you celebrate? We’ll be looking forward to seeing your pictures on social media or in the comments!
