Winter is a time for removing snow, and one fail safe go to is ice. Every winter the road crews bring out the ice trucks to help make driving conditions safer. Homeowners, as well, may salt their drives and sidewalks to prevent injury and accidents on their properties. Sure, we could dust the road and sidewalks with sand to help add grit and friction, but it doesn’t get rid of the dangerous ice so the problem isn’t really solved. So, why does salt work for this unique problem? The answer is in thermodynamics and how a solution can change the freezing temperature.
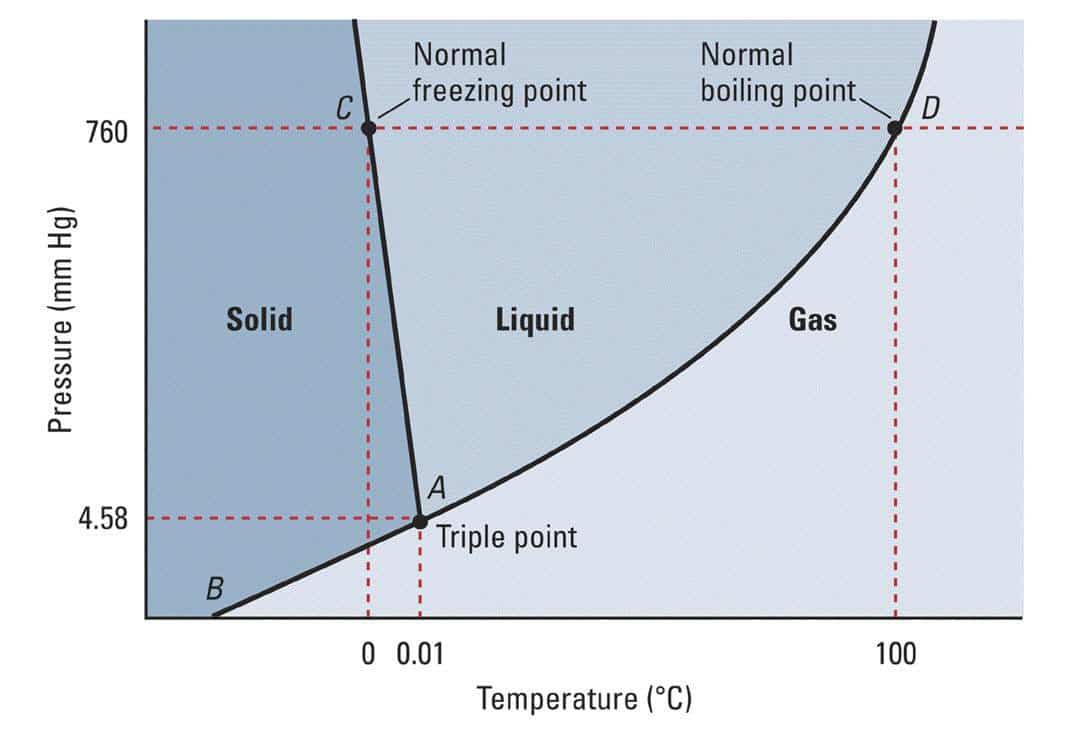
Freezing versus Melting Points Any substance has three phases: solid, liquid, and gas. These phases change with temperature and pressure, which is why water can boil under 100ºC (212ºF) at higher altitudes. The air pressure gets lower as we travel higher, and thus the boiling temperature lowers.
Simplified phase diagram of water. The lines show where the boundary between each phase ranges. At higher pressures, the phases shift to slightly different temperatures. Each substance or solution will have it’s own set of rules that make it useful for predicting and using in labs and beyond. However, when you add something to a solution, it changes to a new set of rules. This is exactly what happens when we add salt to ice: we change it’s phase diagram and set of rules it follows.
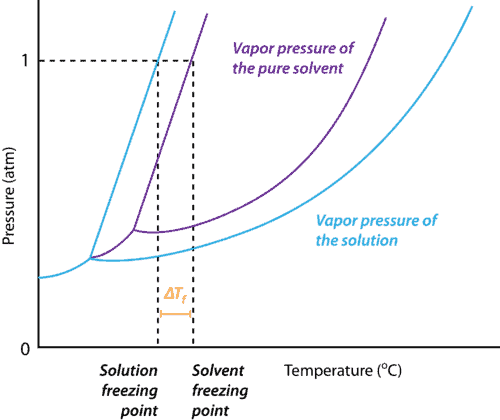
Freezing Point Depression When a solution changes, say from water to salty water, the freezing point changes until the solvent dissolves and washes away. This happens because the solvent is literally different from the original. Take a look at the graph below. The original solvent is shown in purple. It shows that at a certain temperature and pressure the solvent will be a gas, a liquid, or a solid. When any solute is added to it, the solution is fundamentally changed, so it’s graph changes. The blue line represents that change.
The vapor pressure of a solution (blue) is lower than the vapor pressure of a pure solvent (purple). As a result, the freezing point of a solvent decreases when any solute is dissolved into it. Figure from CK-12 Foundation – Christopher Auyeung. Too Cold Is Too Cold Is there such a thing as too cold for rocks? For rock salt to be halite, no, it can’t be too cold. It can, however, be too cold for the solution of water and salt to create the freezing point depression.
Moderation, As Always, Is Key Like all other things in life, you can have too much of a good thing. In this case, the salt leaches into the ground and water. The salt used on roads and sidewalks isn’t purified like the salt we use for food, so sodium, chloride, iron, and other impurities can leach into the ground and water supplies. Research done in New England in the Merrimack River watershed points to an average of 11% of the watersheds having an increased salinity and chlorine amount as a result of the salt leaching into the surrounding areas
Putting it into practice This is a fun party trick that you can use to teach kids about phase diagrams and why salt helps clear the roads of snow. Grab a piece of string, some ice, a glass of water, and some salt. Roll up your sleeves, and enjoy this simple party trick your little ones can show to everyone.
VIDEO
The FREE worksheet for this hands on activity can be found in our digital resource library. Not a member? You can sign up for free here:
Take this to the NEXT level
And get more free resources from our library while you’re at it. Drop your email below to get the depth of learning checklist we use AND a few suggestions on how to focus activities for levels of learning.
Bibliography
{21495:F3XPNEVI}
apa
default
asc
0
11469
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3Afalse%2C%22meta%22%3A%7B%22request_last%22%3A550%2C%22request_next%22%3A50%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22BR2TLM6G%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22National%20Research%20Council%20%28U.S.%29%22%2C%22parsedDate%22%3A%222012%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BNational%20Research%20Council%20%28U.S.%29.%20%282012%29.%20%26lt%3Bi%26gt%3BA%20framework%20for%20K-12%20science%20education%3A%20practices%2C%20crosscutting%20concepts%2C%20and%20core%20ideas%26lt%3B%5C%2Fi%26gt%3B.%20The%20National%20Academies%20Press.%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22book%22%2C%22title%22%3A%22A%20framework%20for%20K-12%20science%20education%3A%20practices%2C%20crosscutting%20concepts%2C%20and%20core%20ideas%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22National%20Research%20Council%20%28U.S.%29%22%7D%5D%2C%22abstractNote%22%3A%22%26quot%3BA%20Framework%20of%20K-12%20Science%20Education%20Standards%20represents%20the%20first%20step%20in%20a%20process%20to%20create%20new%20standards%20in%20K-12%20science%20education%26quot%3B--Foreword%22%2C%22date%22%3A%222012%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%229780309217422%209780309214421%209781280676246%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22eng%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-12T18%3A12%3A51Z%22%7D%7D%2C%7B%22key%22%3A%223CTJZUGM%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Freeman%20et%20al.%22%2C%22parsedDate%22%3A%222014-06-10%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BFreeman%2C%20S.%2C%20Eddy%2C%20S.%20L.%2C%20McDonough%2C%20M.%2C%20Smith%2C%20M.%20K.%2C%20Okoroafor%2C%20N.%2C%20Jordt%2C%20H.%2C%20%26amp%3B%20Wenderoth%2C%20M.%20P.%20%282014%29.%20Active%20learning%20increases%20student%20performance%20in%20science%2C%20engineering%2C%20and%20mathematics.%20%26lt%3Bi%26gt%3BProceedings%20of%20the%20National%20Academy%20of%20Sciences%26lt%3B%5C%2Fi%26gt%3B%2C%20%26lt%3Bi%26gt%3B111%26lt%3B%5C%2Fi%26gt%3B%2823%29%2C%208410%26%23x2013%3B8415.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-DOIURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1073%5C%2Fpnas.1319030111%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1073%5C%2Fpnas.1319030111%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Active%20learning%20increases%20student%20performance%20in%20science%2C%20engineering%2C%20and%20mathematics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Scott%22%2C%22lastName%22%3A%22Freeman%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sarah%20L.%22%2C%22lastName%22%3A%22Eddy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Miles%22%2C%22lastName%22%3A%22McDonough%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michelle%20K.%22%2C%22lastName%22%3A%22Smith%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nnadozie%22%2C%22lastName%22%3A%22Okoroafor%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hannah%22%2C%22lastName%22%3A%22Jordt%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mary%20Pat%22%2C%22lastName%22%3A%22Wenderoth%22%7D%5D%2C%22abstractNote%22%3A%22Significance%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20The%20President%5Cu2019s%20Council%20of%20Advisors%20on%20Science%20and%20Technology%20has%20called%20for%20a%2033%25%20increase%20in%20the%20number%20of%20science%2C%20technology%2C%20engineering%2C%20and%20mathematics%20%28STEM%29%20bachelor%5Cu2019s%20degrees%20completed%20per%20year%20and%20recommended%20adoption%20of%20empirically%20validated%20teaching%20practices%20as%20critical%20to%20achieving%20that%20goal.%20The%20studies%20analyzed%20here%20document%20that%20active%20learning%20leads%20to%20increases%20in%20examination%20performance%20that%20would%20raise%20average%20grades%20by%20a%20half%20a%20letter%2C%20and%20that%20failure%20rates%20under%20traditional%20lecturing%20increase%20by%2055%25%20over%20the%20rates%20observed%20under%20active%20learning.%20The%20analysis%20supports%20theory%20claiming%20that%20calls%20to%20increase%20the%20number%20of%20students%20receiving%20STEM%20degrees%20could%20be%20answered%2C%20at%20least%20in%20part%2C%20by%20abandoning%20traditional%20lecturing%20in%20favor%20of%20active%20learning.%20%5Cn%20%20%20%20%20%20%20%20%20%20%2C%20%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20To%20test%20the%20hypothesis%20that%20lecturing%20maximizes%20learning%20and%20course%20performance%2C%20we%20metaanalyzed%20225%20studies%20that%20reported%20data%20on%20examination%20scores%20or%20failure%20rates%20when%20comparing%20student%20performance%20in%20undergraduate%20science%2C%20technology%2C%20engineering%2C%20and%20mathematics%20%28STEM%29%20courses%20under%20traditional%20lecturing%20versus%20active%20learning.%20The%20effect%20sizes%20indicate%20that%20on%20average%2C%20student%20performance%20on%20examinations%20and%20concept%20inventories%20increased%20by%200.47%20SDs%20under%20active%20learning%20%28%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20n%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20%3D%20158%20studies%29%2C%20and%20that%20the%20odds%20ratio%20for%20failing%20was%201.95%20under%20traditional%20lecturing%20%28%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20n%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20%3D%2067%20studies%29.%20These%20results%20indicate%20that%20average%20examination%20scores%20improved%20by%20about%206%25%20in%20active%20learning%20sections%2C%20and%20that%20students%20in%20classes%20with%20traditional%20lecturing%20were%201.5%20times%20more%20likely%20to%20fail%20than%20were%20students%20in%20classes%20with%20active%20learning.%20Heterogeneity%20analyses%20indicated%20that%20both%20results%20hold%20across%20the%20STEM%20disciplines%2C%20that%20active%20learning%20increases%20scores%20on%20concept%20inventories%20more%20than%20on%20course%20examinations%2C%20and%20that%20active%20learning%20appears%20effective%20across%20all%20class%20sizes%5Cu2014although%20the%20greatest%20effects%20are%20in%20small%20%28%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20n%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20%5Cu2264%2050%29%20classes.%20Trim%20and%20fill%20analyses%20and%20fail-safe%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20n%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20calculations%20suggest%20that%20the%20results%20are%20not%20due%20to%20publication%20bias.%20The%20results%20also%20appear%20robust%20to%20variation%20in%20the%20methodological%20rigor%20of%20the%20included%20studies%2C%20based%20on%20the%20quality%20of%20controls%20over%20student%20quality%20and%20instructor%20identity.%20This%20is%20the%20largest%20and%20most%20comprehensive%20metaanalysis%20of%20undergraduate%20STEM%20education%20published%20to%20date.%20The%20results%20raise%20questions%20about%20the%20continued%20use%20of%20traditional%20lecturing%20as%20a%20control%20in%20research%20studies%2C%20and%20support%20active%20learning%20as%20the%20preferred%2C%20empirically%20validated%20teaching%20practice%20in%20regular%20classrooms.%22%2C%22date%22%3A%222014-06-10%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1073%5C%2Fpnas.1319030111%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fpnas.org%5C%2Fdoi%5C%2Ffull%5C%2F10.1073%5C%2Fpnas.1319030111%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220027-8424%2C%201091-6490%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-12T18%3A09%3A31Z%22%7D%7D%2C%7B%22key%22%3A%22BG6GHXBZ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Jimenez%20et%20al.%22%2C%22parsedDate%22%3A%222021%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BJimenez%2C%20B.%20A.%2C%20Croft%2C%20G.%2C%20Twine%2C%20J.%2C%20%26amp%3B%20Gorey%2C%20J.%20%282021%29.%20Development%20of%20Engineering%20Habits%20of%20Mind%20for%20Students%20With%20Intellectual%20Disability.%20%26lt%3Bi%26gt%3BThe%20Journal%20of%20Special%20Education%26lt%3B%5C%2Fi%26gt%3B%2C%20%26lt%3Bi%26gt%3B55%26lt%3B%5C%2Fi%26gt%3B%283%29%2C%20174%26%23x2013%3B185.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-DOIURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1177%5C%2F00224669211009960%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1177%5C%2F00224669211009960%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Development%20of%20Engineering%20Habits%20of%20Mind%20for%20Students%20With%20Intellectual%20Disability%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Bree%20A.%22%2C%22lastName%22%3A%22Jimenez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gemma%22%2C%22lastName%22%3A%22Croft%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jennifer%22%2C%22lastName%22%3A%22Twine%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jacqueline%22%2C%22lastName%22%3A%22Gorey%22%7D%5D%2C%22abstractNote%22%3A%22The%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20Next%20Generation%20Science%20Standards%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20framework%20outlines%20scientific%20and%20engineering%20practices%20as%20a%20key%20element%20of%20student%20development.%20Educators%20are%20just%20beginning%20to%20discover%20effective%20and%20meaningful%20ways%20to%20teach%20science%20content%20to%20students%20with%20intellectual%20disability%3B%20however%2C%20the%20literature%20on%20teaching%20science%20practices%20is%20still%20limited%20and%20engineering%20practices%20nonexistent.%20This%20study%20utilized%20a%20quasi-experimental%20group%20design%20to%20investigate%20the%20impact%20of%20universally%20designed%20engineering%20curriculum%20on%2043%20students%20with%20intellectual%20disability%20and%20autism%20display%20of%20engineering%20habits%20of%20mind%20%28e.g.%2C%20persist%20and%20learn%20from%20failure%29%20during%20ongoing%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20Engineering%20is%20Elementary%20%5Cn%20%20%20%20%20%20%20%20%20%20%20%20%20%20design%20challenges.%20A%20statistically%20significant%20difference%20between%20the%20control%20and%20treatment%20groups%20was%20found.%20Findings%20suggest%20engineering%20instruction%20may%20support%20student%20development%20of%20problem-solving%20skills.%22%2C%22date%22%3A%2211%5C%2F2021%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1177%5C%2F00224669211009960%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fjournals.sagepub.com%5C%2Fdoi%5C%2F10.1177%5C%2F00224669211009960%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220022-4669%2C%201538-4764%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-09T17%3A53%3A37Z%22%7D%7D%2C%7B%22key%22%3A%22VPT8UBHX%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Centre%20for%20Real-World%20Learning%22%2C%22parsedDate%22%3A%222014-05%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BCentre%20for%20Real-World%20Learning.%20%282014%2C%20May%29.%20%26lt%3Bi%26gt%3BThinking%20like%20an%20engineer%5CnImplications%20for%20the%5Cneducation%20system%26lt%3B%5C%2Fi%26gt%3B.%20Royal%20Academy%20of%20Engineering.%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22document%22%2C%22title%22%3A%22Thinking%20like%20an%20engineer%5CnImplications%20for%20the%5Cneducation%20system%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22Centre%20for%20Real-World%20Learning%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22type%22%3A%22%22%2C%22date%22%3A%222014-05%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-09T17%3A45%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22JZDI8S5D%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22parsedDate%22%3A%222021-11-10%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BCochrane%2C%20P.%20%282021%2C%20November%2010%29.%20%26lt%3Bi%26gt%3BPrivate%20Wells%20%26amp%3B%20Testing%20Drinking%20Water%26lt%3B%5C%2Fi%26gt%3B%20%5BVideo%20recording%5D.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fyoutu.be%5C%2F9mtMqsTiNag%3Fsi%3Do2ax8328ItI-2GNu%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fyoutu.be%5C%2F9mtMqsTiNag%3Fsi%3Do2ax8328ItI-2GNu%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22videoRecording%22%2C%22title%22%3A%22Private%20Wells%20%26%20Testing%20Drinking%20Water%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22creator%22%2C%22firstName%22%3A%22Paul%22%2C%22lastName%22%3A%22Cochrane%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22videoRecordingFormat%22%3A%22%22%2C%22studio%22%3A%22%22%2C%22date%22%3A%222021-11-10%22%2C%22runningTime%22%3A%22%22%2C%22ISBN%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fyoutu.be%5C%2F9mtMqsTiNag%3Fsi%3Do2ax8328ItI-2GNu%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-06T21%3A08%3A31Z%22%7D%7D%2C%7B%22key%22%3A%22CD2UPCAC%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22NRCS%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BNRCS.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BSoil%20Testing%20Small%20Scale%20Solutions%20for%20Your%20Farm%26lt%3B%5C%2Fi%26gt%3B.%20USDA.%20Retrieved%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.nrcs.usda.gov%5C%2Fsites%5C%2Fdefault%5C%2Ffiles%5C%2F2023-01%5C%2FSoil%2520Testing-%2520Small%2520Scale%2520Solutions%2520for%2520your%2520Farm.pdf%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.nrcs.usda.gov%5C%2Fsites%5C%2Fdefault%5C%2Ffiles%5C%2F2023-01%5C%2FSoil%2520Testing-%2520Small%2520Scale%2520Solutions%2520for%2520your%2520Farm.pdf%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22document%22%2C%22title%22%3A%22Soil%20Testing%20Small%20Scale%20Solutions%20for%20Your%20Farm%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22NRCS%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22type%22%3A%22%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.nrcs.usda.gov%5C%2Fsites%5C%2Fdefault%5C%2Ffiles%5C%2F2023-01%5C%2FSoil%2520Testing-%2520Small%2520Scale%2520Solutions%2520for%2520your%2520Farm.pdf%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T22%3A19%3A46Z%22%7D%7D%2C%7B%22key%22%3A%222WWWRCA4%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22NRCS%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BNRCS.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BSoil%20Testing%20for%20Small%20Farms%20and%20Gardens%26lt%3B%5C%2Fi%26gt%3B%20%5BTechnical%20Notes%5D.%20USDA.%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22document%22%2C%22title%22%3A%22Soil%20Testing%20for%20Small%20Farms%20and%20Gardens%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22NRCS%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22type%22%3A%22Technical%20Notes%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T22%3A06%3A38Z%22%7D%7D%2C%7B%22key%22%3A%22E8DJ2SKB%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Paul%20Cochrane%22%2C%22parsedDate%22%3A%222021-11-10%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BPaul%20Cochrane.%20%282021%2C%20November%2010%29.%20%26lt%3Bi%26gt%3BPrivate%20Wells%20%26amp%3B%20Testing%20Drinking%20Water%26lt%3B%5C%2Fi%26gt%3B%20%5BVideo%20recording%5D.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.youtube.com%5C%2Fwatch%3Fv%3D9mtMqsTiNag%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.youtube.com%5C%2Fwatch%3Fv%3D9mtMqsTiNag%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22videoRecording%22%2C%22title%22%3A%22Private%20Wells%20%26%20Testing%20Drinking%20Water%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22director%22%2C%22name%22%3A%22Paul%20Cochrane%22%7D%5D%2C%22abstractNote%22%3A%22The%20U.S.%20Environmental%20Protection%20Agency%20%28EPA%29%20estimates%20that%20more%20than%2013%20million%20households%20in%20the%20United%20States%20rely%20on%20private%20wells%20for%20drinking%20water%20%28US%20Census%20American%20Housing%20Survey%202017%29.%20%5Cn%5CnThe%20agency%20does%20not%20regulate%20private%20wells%20nor%20does%20it%20provide%20recommended%20criteria%20or%20standards%20for%20individual%20wells.%20Private%20well%20owners%20are%20responsible%20for%20the%20safety%20of%20their%20water%2C%20but%20EPA%20does%20offer%20inf%22%2C%22videoRecordingFormat%22%3A%22%22%2C%22studio%22%3A%22%22%2C%22date%22%3A%222021-11-10%22%2C%22runningTime%22%3A%2203%3A38%22%2C%22ISBN%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.youtube.com%5C%2Fwatch%3Fv%3D9mtMqsTiNag%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T20%3A09%3A20Z%22%7D%7D%2C%7B%22key%22%3A%228DWR739X%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22US%20EPA%22%2C%22parsedDate%22%3A%222015-05-06%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BUS%20EPA%2C%20O.%20%282015%2C%20May%206%29.%20%26lt%3Bi%26gt%3BProtect%20Your%20Home%26%23x2019%3Bs%20Water%26lt%3B%5C%2Fi%26gt%3B%20%5BOverviews%20and%20Factsheets%5D.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.epa.gov%5C%2Fprivatewells%5C%2Fprotect-your-homes-water%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.epa.gov%5C%2Fprivatewells%5C%2Fprotect-your-homes-water%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Protect%20Your%20Home%27s%20Water%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22OW%22%2C%22lastName%22%3A%22US%20EPA%22%7D%5D%2C%22abstractNote%22%3A%22This%20page%20informs%20private%20drinking%20water%20well%20owners%20of%20the%20importance%20of%20testing%2C%20post-flood%20actions%2C%20and%20preventative%20measures%20to%20ensure%20well%20water%20quality.%22%2C%22date%22%3A%222015-05-06T12%3A38%3A06-04%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.epa.gov%5C%2Fprivatewells%5C%2Fprotect-your-homes-water%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T20%3A09%3A07Z%22%7D%7D%2C%7B%22key%22%3A%22JWKQARX9%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Douglas%22%2C%22parsedDate%22%3A%222021-05-11%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BDouglas%2C%20R.%20%282021%2C%20May%2011%29.%20%26lt%3Bi%26gt%3BSoil%20Test%20Information%20%7C%20N.C.%20Cooperative%20Extension%26lt%3B%5C%2Fi%26gt%3B.%20Haywood%20County%20Center.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fhaywood.ces.ncsu.edu%5C%2Fsoil-test-information%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fhaywood.ces.ncsu.edu%5C%2Fsoil-test-information%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Soil%20Test%20Information%20%7C%20N.C.%20Cooperative%20Extension%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rachel%22%2C%22lastName%22%3A%22Douglas%22%7D%5D%2C%22abstractNote%22%3A%22Don%5Cu2019t%20guess%2C%20soil%20test.Any%20successful%20gardener%20in%20this%20area%20will%20tell%20you%20that%20the%20key%20to%20a%20beautiful%20lawn%20or%20garden%20starts%20with%20healthy%20soil.%20The%20only%20way%20to%20ensure%20your%20soil%20is%20healthy%2C%20however%2C%20is%5Cu2026%22%2C%22date%22%3A%222021-05-11T08%3A49%3A46-04%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fhaywood.ces.ncsu.edu%5C%2Fsoil-test-information%5C%2F%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T20%3A08%3A59Z%22%7D%7D%2C%7B%22key%22%3A%22TUM3JXFF%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Jones%22%2C%22parsedDate%22%3A%222014-08-20%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BJones%2C%20M.%20%282014%2C%20August%2020%29.%20%26lt%3Bi%26gt%3BSoil%20Testing%20for%20Lawns%20and%20Gardens%20%7C%20N.C.%20Cooperative%20Extension%26lt%3B%5C%2Fi%26gt%3B.%20Chatham%20County%20Center.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fchatham.ces.ncsu.edu%5C%2Fsoil-testing-for-lawns-and-gardens%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fchatham.ces.ncsu.edu%5C%2Fsoil-testing-for-lawns-and-gardens%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Soil%20Testing%20for%20Lawns%20and%20Gardens%20%7C%20N.C.%20Cooperative%20Extension%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Matt%22%2C%22lastName%22%3A%22Jones%22%7D%5D%2C%22abstractNote%22%3A%22From%20April%20-%20November%2C%20Have%20Your%20Soil%20Tested%20At%20No%20Charge%20For%20questions%2C%20contact%20Matt%20Jones%20%28matt_jones%40ncsu.edu%29%20Learn%20how%20to%20pick%20up%20soil%20boxes%20and%20drop%20off%20soil%20samples%20for%20delivery%20here.%20Review%5Cu2026%22%2C%22date%22%3A%222014-08-20T07%3A09%3A13-04%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fchatham.ces.ncsu.edu%5C%2Fsoil-testing-for-lawns-and-gardens%5C%2F%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T20%3A08%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22E9XWHRVT%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BA%20Gardener%26%23x2019%3Bs%20Guide%20to%20Soil%20Testing%20%7C%20NC%20State%20Extension%20Publications%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20Retrieved%20March%202%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fcontent.ces.ncsu.edu%5C%2Fa-gardeners-guide-to-soil-testing%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fcontent.ces.ncsu.edu%5C%2Fa-gardeners-guide-to-soil-testing%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22A%20Gardener%27s%20Guide%20to%20Soil%20Testing%20%7C%20NC%20State%20Extension%20Publications%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22This%20publication%20tells%20gardeners%20why%20they%20should%20test%20their%20soil%2C%20how%20to%20obtain%20a%20soil%20test%20and%20interpret%20the%20results%20and%20how%20to%20use%20the%20soil%20test%20to%20improve%20their%20soils.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fcontent.ces.ncsu.edu%5C%2Fa-gardeners-guide-to-soil-testing%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T20%3A08%3A52Z%22%7D%7D%2C%7B%22key%22%3A%22JMXJF2VJ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BSoil%20Testing%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20Purdue%20University%20-%20Extension.%20Retrieved%20March%202%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fextension.purdue.edu%5C%2Fcounty%5C%2Fmarion%5C%2Fanr%5C%2Fsoil-testing.html%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fextension.purdue.edu%5C%2Fcounty%5C%2Fmarion%5C%2Fanr%5C%2Fsoil-testing.html%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Soil%20Testing%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22Collecting%20Soil%20Samples%20for%20Testing%2C%20Soil%20Testing%20Info%2C%20Soil%20Testing%20Labs%2C%20Soil%20pH%2C%20Soil%20testing%3A%20heavy%20metals.%20To%20grow%20good%20plants%2C%20you%20need%20good%20soil.%20The%20only%20way%20to%20tell%20what%20your%20soil%20really%20needs%20is%20to%20take%20a%20soil%20test.%20Applying%20too%20much%20fertilizer%20could%20be%20detrimental%20to%20your%20plants.%20You%20could%20waste%20money%20or%20pollute%20the%20environment.%20Adding%20too%20little%20fertilizer%20or%20the%20wrong%20fertilizer%20could%20produce%20little%20or%20no%20results.%20Therefore%2C%20for%20optimum%20plant%20growth%2C%20it%20is%20highly%20recommended%20to%20test%20your%20soil%20pH%20and%20nutrient%20status%20every%203%20to%205%20years.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fextension.purdue.edu%5C%2Fcounty%5C%2Fmarion%5C%2Fanr%5C%2Fsoil-testing.html%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T20%3A06%3A48Z%22%7D%7D%2C%7B%22key%22%3A%22EGV55KV6%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22parsedDate%22%3A%222026-03-08%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BProton%20Mail%26lt%3B%5C%2Fi%26gt%3B.%20%282026%2C%20March%208%29.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fmail.proton.me%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fmail.proton.me%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Proton%20Mail%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22Proton%20Mail%20is%20based%20in%20Switzerland%20and%20uses%20advanced%20encryption%20to%20keep%20your%20data%20safe.%20Apps%20available%20for%20Android%2C%20iOS%2C%20and%20desktop%20devices.%22%2C%22date%22%3A%222026-03-08%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fmail.proton.me%5C%2F%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-03-02T19%3A12%3A37Z%22%7D%7D%2C%7B%22key%22%3A%229F28B8Q3%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22anubha%22%2C%22parsedDate%22%3A%222024-11-11%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3Banubha.%20%282024%2C%20November%2011%29.%20Lab%20Work%20Made%20Easy%3A%20Choosing%20the%20Right%20Chemistry%20Glassware.%20%26lt%3Bi%26gt%3BScientific%20Lab%20Equipment%20Manufacturer%20and%20Supplier%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.shivsons.com%5C%2Flab-work-made-easy-choosing-the-right-chemistry-glassware%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.shivsons.com%5C%2Flab-work-made-easy-choosing-the-right-chemistry-glassware%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Lab%20Work%20Made%20Easy%3A%20Choosing%20the%20Right%20Chemistry%20Glassware%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22anubha%22%7D%5D%2C%22abstractNote%22%3A%22Find%20the%20right%20chemistry%20glassware%20for%20your%20lab.%20Simplify%20experiments%20and%20boost%20accuracy%20with%20essential%20tips%20for%20lab%20work%20made%20easy.%22%2C%22blogTitle%22%3A%22Scientific%20Lab%20Equipment%20Manufacturer%20and%20Supplier%22%2C%22date%22%3A%222024-11-11T04%3A55%3A33%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.shivsons.com%5C%2Flab-work-made-easy-choosing-the-right-chemistry-glassware%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-26T17%3A39%3A32Z%22%7D%7D%2C%7B%22key%22%3A%225E8EW9NC%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22The4%22%2C%22parsedDate%22%3A%222026-01-29%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BThe4.%20%282026%2C%20January%2029%29.%20%26lt%3Bi%26gt%3BA%20list%20of%20glassware%20equipment%20for%20high%20school%20chemistry%20lab.%20-%20Science%26lt%3B%5C%2Fi%26gt%3B.%20Science%20Equip.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.scienceequip.com.au%5C%2Fblogs%5C%2Fnews%5C%2Fa-list-of-glassware-equipment-for-high-school-chemistry-lab%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.scienceequip.com.au%5C%2Fblogs%5C%2Fnews%5C%2Fa-list-of-glassware-equipment-for-high-school-chemistry-lab%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22A%20list%20of%20glassware%20equipment%20for%20high%20school%20chemistry%20lab.%20-%20Science%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22The4%22%7D%5D%2C%22abstractNote%22%3A%22In%20the%20article%20we%20take%20a%20closer%20look%20at%20some%20of%20the%20most%20common%20glassware%20equipment%20utilised%20by%20high%20school%20chemistry%20lab%20today.%22%2C%22date%22%3A%222026-01-29%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.scienceequip.com.au%5C%2Fblogs%5C%2Fnews%5C%2Fa-list-of-glassware-equipment-for-high-school-chemistry-lab%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-26T17%3A39%3A24Z%22%7D%7D%2C%7B%22key%22%3A%22KSL995K3%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22says%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3Bsays%2C%20G.%20johnson.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BBeakers%20vs.%20Flasks%3A%20The%20Pros%20and%20Cons%20of%20Frequently%20Used%20Lab%20Glassware%26lt%3B%5C%2Fi%26gt%3B.%20Lab%20Pro%20Inc.%20Retrieved%20February%2026%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Flabproinc.com%5C%2Fblogs%5C%2Flaboratory-equipment%5C%2Fbeakers-vs-flasks-the-pros-and-cons-of-frequently-used-lab-glassware%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Flabproinc.com%5C%2Fblogs%5C%2Flaboratory-equipment%5C%2Fbeakers-vs-flasks-the-pros-and-cons-of-frequently-used-lab-glassware%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Beakers%20vs.%20Flasks%3A%20The%20Pros%20and%20Cons%20of%20Frequently%20Used%20Lab%20Glassware%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gary%20johnson%22%2C%22lastName%22%3A%22says%22%7D%5D%2C%22abstractNote%22%3A%22Find%20out%20%5Cu201cwhat%20is%20a%20beaker%20and%20a%20flask%5Cu201d%2C%20%5Cu201cwhat%20are%20they%20used%20for%20in%20labs%5Cu201d.%20Compare%20beakers%20and%20flasks%20to%20choose%20the%20right%20one%20for%20precision%20and%20consistency.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Flabproinc.com%5C%2Fblogs%5C%2Flaboratory-equipment%5C%2Fbeakers-vs-flasks-the-pros-and-cons-of-frequently-used-lab-glassware%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-26T17%3A16%3A01Z%22%7D%7D%2C%7B%22key%22%3A%22FHRVBIGC%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BUnderstanding%20Laboratory%20Glassware%20Classifications%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20Globe%20Scientific.%20Retrieved%20February%2026%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fglobescientific.com%5C%2Fblogs%5C%2Fblog%5C%2Funderstanding-laboratory-glassware-classifications%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fglobescientific.com%5C%2Fblogs%5C%2Fblog%5C%2Funderstanding-laboratory-glassware-classifications%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Understanding%20Laboratory%20Glassware%20Classifications%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22As%20a%20busy%20lab%20professional%2C%20you%20need%20to%20have%20confidence%20that%20the%20glassware%20in%20your%20lab%20is%20reliable%20and%20of%20high%20quality.%20You%20don%5Cu2019t%20have%20time%20to%20worry%20about%20how%20your%20glassware%20will%20perform%20in%20measurement%20settings%5Cu2014you%20need%20the%20peace%20of%20mind%20that%20it%20can%20compete%20with%20the%20best%20products%20on%20the%20market.%20Luckily%2C%20product%20manufac%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fglobescientific.com%5C%2Fblogs%5C%2Fblog%5C%2Funderstanding-laboratory-glassware-classifications%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-26T17%3A09%3A58Z%22%7D%7D%2C%7B%22key%22%3A%224ET35UTR%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BLaboratory%20Beakers%20Guide%20%26%23x2013%3B%20Types%20%26amp%3B%20Uses%20Explained%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20ChemScience%20Inc.%20Retrieved%20February%2026%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.chemscience.com%5C%2Fblog%5C%2Fwww.chemscience.com%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.chemscience.com%5C%2Fblog%5C%2Fwww.chemscience.com%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Laboratory%20Beakers%20Guide%20%5Cu2013%20Types%20%26%20Uses%20Explained%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22Explore%20different%20beaker%20types%20glass%2C%20plastic%2C%20metal%20for%20accurate%20lab%20use.%20Find%20tips%20for%20safe%20handling%20and%20choosing%20the%20right%20container%20for%20better%20accuracy%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.chemscience.com%5C%2Fblog%5C%2Fwww.chemscience.com%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-26T17%3A09%3A20Z%22%7D%7D%2C%7B%22key%22%3A%22NK7965IH%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Carolina.Biological%22%2C%22parsedDate%22%3A%222020-01-01%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BCarolina.Biological.%20%282020%2C%20January%201%29.%20%26lt%3Bi%26gt%3BStocking%20a%20laboratory%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fknowledge.carolina.com%5C%2Fproduct-resources%5C%2Fbuying-guides%5C%2Fultimate-lab-basics-buying-guide%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fknowledge.carolina.com%5C%2Fproduct-resources%5C%2Fbuying-guides%5C%2Fultimate-lab-basics-buying-guide%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Stocking%20a%20laboratory%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22Carolina.Biological%22%7D%5D%2C%22abstractNote%22%3A%22Start%20the%20new%20school%20year%20off%20right%20by%20stocking%20up%20on%20top-brand%20supplies%20from%20a%20name%20you%20can%20trust.%22%2C%22blogTitle%22%3A%22%22%2C%22date%22%3A%222020-01-01T20%3A26%3A23-05%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fknowledge.carolina.com%5C%2Fproduct-resources%5C%2Fbuying-guides%5C%2Fultimate-lab-basics-buying-guide%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-26T17%3A02%3A58Z%22%7D%7D%2C%7B%22key%22%3A%2282RD8EEB%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lam%20et%20al.%22%2C%22parsedDate%22%3A%222024-05-21%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BLam%2C%20K.%2C%20Armstrong%2C%20B.%2C%20%26amp%3B%20Margelefsky%2C%20E.%20L.%20%282024%29.%20Correction%20to%20%26%23x201C%3BGravimetric%20and%20Thermal-Imaging%20Characterization%20of%20Water-Free%20Reflux%20Condensers%20for%20Laboratory%20Applications.%26%23x201D%3B%20%26lt%3Bi%26gt%3BACS%20Omega%26lt%3B%5C%2Fi%26gt%3B%2C%20%26lt%3Bi%26gt%3B9%26lt%3B%5C%2Fi%26gt%3B%2820%29%2C%2022508.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-DOIURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsomega.4c03508%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsomega.4c03508%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Correction%20to%20%5C%22Gravimetric%20and%20Thermal-Imaging%20Characterization%20of%20Water-Free%20Reflux%20Condensers%20for%20Laboratory%20Applications%5C%22%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kevin%22%2C%22lastName%22%3A%22Lam%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Brittany%22%2C%22lastName%22%3A%22Armstrong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eric%20L.%22%2C%22lastName%22%3A%22Margelefsky%22%7D%5D%2C%22abstractNote%22%3A%22%5BThis%20corrects%20the%20article%20DOI%3A%2010.1021%5C%2Facsomega.4c00411.%5D.%22%2C%22date%22%3A%222024-05-21%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facsomega.4c03508%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222470-1343%22%2C%22language%22%3A%22eng%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T20%3A03%3A14Z%22%7D%7D%2C%7B%22key%22%3A%22F4YXNT8D%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lam%20et%20al.%22%2C%22parsedDate%22%3A%222024-04-09%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BLam%2C%20K.%2C%20Armstrong%2C%20B.%2C%20%26amp%3B%20Margelefsky%2C%20E.%20L.%20%282024%29.%20Gravimetric%20and%20Thermal-Imaging%20Characterization%20of%20Water-Free%20Reflux%20Condensers%20for%20Laboratory%20Applications.%20%26lt%3Bi%26gt%3BACS%20Omega%26lt%3B%5C%2Fi%26gt%3B%2C%20%26lt%3Bi%26gt%3B9%26lt%3B%5C%2Fi%26gt%3B%2814%29%2C%2016563%26%23x2013%3B16571.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-DOIURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsomega.4c00411%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1021%5C%2Facsomega.4c00411%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Gravimetric%20and%20Thermal-Imaging%20Characterization%20of%20Water-Free%20Reflux%20Condensers%20for%20Laboratory%20Applications%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kevin%22%2C%22lastName%22%3A%22Lam%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Brittany%22%2C%22lastName%22%3A%22Armstrong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eric%20L.%22%2C%22lastName%22%3A%22Margelefsky%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222024-04-09%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1021%5C%2Facsomega.4c00411%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fpubs.acs.org%5C%2Fdoi%5C%2F10.1021%5C%2Facsomega.4c00411%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%222470-1343%2C%202470-1343%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T20%3A02%3A52Z%22%7D%7D%2C%7B%22key%22%3A%228G9YUDWQ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Fordham%22%2C%22parsedDate%22%3A%222024-02-07%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BFordham%2C%20C.%20%282024%2C%20February%207%29.%20Condenser%20Chemistry%3A%20What%26%23x2019%3Bs%20the%20Difference%20Between%20Distillation%20%26amp%3B%20Reflux%3F%20%26lt%3Bi%26gt%3BAsynt%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.asynt.com%5C%2Fblog%5C%2Fcondenser-chemistry-difference-between-distillation-reflux%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.asynt.com%5C%2Fblog%5C%2Fcondenser-chemistry-difference-between-distillation-reflux%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Condenser%20Chemistry%3A%20What%27s%20the%20Difference%20Between%20Distillation%20%26%20Reflux%3F%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cameron%22%2C%22lastName%22%3A%22Fordham%22%7D%5D%2C%22abstractNote%22%3A%22Explore%20the%20differences%20between%20distillation%20and%20reflux%20in%20condenser%20chemistry%2C%20from%20simple%20to%20vacuum%20distillation%20and%20their%20key%20applications.%22%2C%22blogTitle%22%3A%22Asynt%22%2C%22date%22%3A%222024-02-07T12%3A41%3A42%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.asynt.com%5C%2Fblog%5C%2Fcondenser-chemistry-difference-between-distillation-reflux%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-GB%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T19%3A36%3A52Z%22%7D%7D%2C%7B%22key%22%3A%227QMSRV59%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Fordham%22%2C%22parsedDate%22%3A%222024-02-07%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BFordham%2C%20C.%20%282024%2C%20February%207%29.%20Condenser%20Chemistry%3A%20What%26%23x2019%3Bs%20the%20Difference%20Between%20Distillation%20%26amp%3B%20Reflux%3F%20%26lt%3Bi%26gt%3BAsynt%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.asynt.com%5C%2Fblog%5C%2Fcondenser-chemistry-difference-between-distillation-reflux%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.asynt.com%5C%2Fblog%5C%2Fcondenser-chemistry-difference-between-distillation-reflux%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Condenser%20Chemistry%3A%20What%27s%20the%20Difference%20Between%20Distillation%20%26%20Reflux%3F%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cameron%22%2C%22lastName%22%3A%22Fordham%22%7D%5D%2C%22abstractNote%22%3A%22Explore%20the%20differences%20between%20distillation%20and%20reflux%20in%20condenser%20chemistry%2C%20from%20simple%20to%20vacuum%20distillation%20and%20their%20key%20applications.%22%2C%22blogTitle%22%3A%22Asynt%22%2C%22date%22%3A%222024-02-07T12%3A41%3A42%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.asynt.com%5C%2Fblog%5C%2Fcondenser-chemistry-difference-between-distillation-reflux%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-GB%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T19%3A23%3A25Z%22%7D%7D%2C%7B%22key%22%3A%229QSHPRKP%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22admin%22%2C%22parsedDate%22%3A%222025-10-28%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3Badmin.%20%282025%2C%20October%2028%29.%20What%20Is%20Reflux%20Distillation%3F%20A%20Complete%20Guide.%20%26lt%3Bi%26gt%3BBorosil%20Scientific%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.borosilscientific.com%5C%2Fwhat-is-reflux-distillation-complete-guide%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.borosilscientific.com%5C%2Fwhat-is-reflux-distillation-complete-guide%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22What%20Is%20Reflux%20Distillation%3F%20A%20Complete%20Guide%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22admin%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22blogTitle%22%3A%22Borosil%20Scientific%22%2C%22date%22%3A%222025-10-28T12%3A03%3A55%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.borosilscientific.com%5C%2Fwhat-is-reflux-distillation-complete-guide%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T19%3A22%3A37Z%22%7D%7D%2C%7B%22key%22%3A%22M3IV7IHG%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Reflux%20and%20distillation%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BReflux%20and%20distillation.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BReflux%20and%20distillation%20%26%23x2013%3B%20practical%20videos%20%7C%2016%26%23x2013%3B18%20students%26lt%3B%5C%2Fi%26gt%3B.%20RSC%20Education.%20Retrieved%20February%2018%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fedu.rsc.org%5C%2Fpractical%5C%2Freflux-and-distillation-practical-videos-16-18-students%5C%2F4012294.article%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fedu.rsc.org%5C%2Fpractical%5C%2Freflux-and-distillation-practical-videos-16-18-students%5C%2F4012294.article%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Reflux%20and%20distillation%20%5Cu2013%20practical%20videos%20%7C%2016%5Cu201318%20students%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22Reflux%20and%20distillation%22%7D%5D%2C%22abstractNote%22%3A%22Learn%20how%20to%20carry%20out%20two%20common%20practical%20techniques%2C%20understand%20how%20they%20differ%20and%20their%20applications.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fedu.rsc.org%5C%2Fpractical%5C%2Freflux-and-distillation-practical-videos-16-18-students%5C%2F4012294.article%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T19%3A21%3A05Z%22%7D%7D%2C%7B%22key%22%3A%22BTDJWD9C%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BWater%20flow%20in%20condenser%20-%20ECHEMI.com%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20ECHEMI.%20Retrieved%20February%2018%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.echemi.com%5C%2Fcommunity%5C%2Fwater-flow-in-condenser_mjart2205112471_431.html%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.echemi.com%5C%2Fcommunity%5C%2Fwater-flow-in-condenser_mjart2205112471_431.html%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Water%20flow%20in%20condenser%20-%20ECHEMI.com%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22How%20should%20the%20water%20flow%20in%20your%20condenser%3F%20From%20the%20flask%20to%20the%20top%20of%20the%20condenser%2C%20or%20from%20the%20top%20of%20the%20condenser%20back%20to%20the%20flask%3F%20I%20could%20imagine%20both%3A%20flask%20to%20top%20will%20fill%20the%20entire%20condenser%20with%20water%2C%20because%20of%20the%20resistance%20of%20gravity%2C%20enlarging%20cooling%20area%2C%20or%20top%20to%20flask%2C%20because%20this%20will%20generate%20counter%20current%20exchange%20increasing%20heat%20flow%2C%20but%20then%20it%20is%20almost%20impossible%20to%20get%20a%20decent%20cooling%20surface%20without%20extreme%20water%20pressure.This%20picture%20suggests%20the%20first.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.echemi.com%5C%2Fcommunity%5C%2Fwater-flow-in-condenser_mjart2205112471_431.html%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T18%3A22%3A45Z%22%7D%7D%2C%7B%22key%22%3A%22M5PCE2WR%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Abrahams%20and%20Millar%22%2C%22parsedDate%22%3A%222008-11-17%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BAbrahams%2C%20I.%2C%20%26amp%3B%20Millar%2C%20R.%20%282008%29.%20Does%20Practical%20Work%20Really%20Work%3F%20A%20study%20of%20the%20effectiveness%20of%20practical%20work%20as%20a%20teaching%20and%20learning%20method%20in%20school%20science.%20%26lt%3Bi%26gt%3BInternational%20Journal%20of%20Science%20Education%26lt%3B%5C%2Fi%26gt%3B%2C%20%26lt%3Bi%26gt%3B30%26lt%3B%5C%2Fi%26gt%3B%2814%29%2C%201945%26%23x2013%3B1969.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-DOIURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1080%5C%2F09500690701749305%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1080%5C%2F09500690701749305%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Does%20Practical%20Work%20Really%20Work%3F%20A%20study%20of%20the%20effectiveness%20of%20practical%20work%20as%20a%20teaching%20and%20learning%20method%20in%20school%20science%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ian%22%2C%22lastName%22%3A%22Abrahams%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Robin%22%2C%22lastName%22%3A%22Millar%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222008-11-17%22%2C%22section%22%3A%22%22%2C%22partNumber%22%3A%22%22%2C%22partTitle%22%3A%22%22%2C%22DOI%22%3A%2210.1080%5C%2F09500690701749305%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.tandfonline.com%5C%2Fdoi%5C%2Fabs%5C%2F10.1080%5C%2F09500690701749305%22%2C%22PMID%22%3A%22%22%2C%22PMCID%22%3A%22%22%2C%22ISSN%22%3A%220950-0693%2C%201464-5289%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T18%3A05%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22IIMT5KZS%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Vogel%22%2C%22parsedDate%22%3A%221977%22%2C%22numChildren%22%3A0%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BVogel%2C%20A.%20I.%20%281977%29.%20%26lt%3Bi%26gt%3BA%20text-book%20of%20practical%20organic%20chemistry%3A%20incl.%20qualitative%20organic%20analysis%26lt%3B%5C%2Fi%26gt%3B%20%283.%20ed.%3B%20new%20impr%29.%20Longmans.%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22book%22%2C%22title%22%3A%22A%20text-book%20of%20practical%20organic%20chemistry%3A%20incl.%20qualitative%20organic%20analysis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Arthur%20Israel%22%2C%22lastName%22%3A%22Vogel%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%221977%22%2C%22originalDate%22%3A%22%22%2C%22originalPublisher%22%3A%22%22%2C%22originalPlace%22%3A%22%22%2C%22format%22%3A%22%22%2C%22ISBN%22%3A%229780582442450%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-18T17%3A48%3A38Z%22%7D%7D%2C%7B%22key%22%3A%22YECMMI5M%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Indiana%22%2C%22parsedDate%22%3A%222025-10-22%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BIndiana%2C%20W.%20Q.%20in.%20%282025%2C%20October%2022%29.%20%26lt%3Bi%26gt%3BCommunity%20Drinking%20Water%20Information%20-%20Madison%20County%26lt%3B%5C%2Fi%26gt%3B.%20Water%20Quality%20in%20Indiana.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.in.gov%5C%2Fidem%5C%2Fcleanwater%5C%2Fdrinking-water%5C%2Fcommunity-drinking-water-info-madison-co%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.in.gov%5C%2Fidem%5C%2Fcleanwater%5C%2Fdrinking-water%5C%2Fcommunity-drinking-water-info-madison-co%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Community%20Drinking%20Water%20Information%20-%20Madison%20County%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Water%20Quality%20in%22%2C%22lastName%22%3A%22Indiana%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222025-10-22T15%3A53%3A10-04%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.in.gov%5C%2Fidem%5C%2Fcleanwater%5C%2Fdrinking-water%5C%2Fcommunity-drinking-water-info-madison-co%5C%2F%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-06T21%3A40%3A59Z%22%7D%7D%2C%7B%22key%22%3A%224NDMQFUB%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Schneider%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BSchneider%2C%20K.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BThese%20Indiana%20residents%20have%20brown%20water%20and%20want%20their%20town%20to%20tell%20them%20why%26lt%3B%5C%2Fi%26gt%3B.%20The%20Indianapolis%20Star.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.indystar.com%5C%2Fstory%5C%2Fnews%5C%2Fenvironment%5C%2F2025%5C%2F10%5C%2F06%5C%2Findiana-brown-water-quality-safe-drinking%5C%2F86481832007%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.indystar.com%5C%2Fstory%5C%2Fnews%5C%2Fenvironment%5C%2F2025%5C%2F10%5C%2F06%5C%2Findiana-brown-water-quality-safe-drinking%5C%2F86481832007%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22These%20Indiana%20residents%20have%20brown%20water%20and%20want%20their%20town%20to%20tell%20them%20why%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Karl%22%2C%22lastName%22%3A%22Schneider%22%7D%5D%2C%22abstractNote%22%3A%22Residents%20in%20Lapel%2C%20Indiana%2C%20are%20experiencing%20brown%20water%20coming%20out%20of%20their%20taps%20and%20are%20worried%20that%20the%20water%20is%20not%20safe%20to%20drink.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.indystar.com%5C%2Fstory%5C%2Fnews%5C%2Fenvironment%5C%2F2025%5C%2F10%5C%2F06%5C%2Findiana-brown-water-quality-safe-drinking%5C%2F86481832007%5C%2F%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T20%3A38%3A48Z%22%7D%7D%2C%7B%22key%22%3A%2266VVKVCR%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BFixed%20Costs%20vs%20Variable%20Costs%3A%20A%20Practical%20Guide%20with%20Examples%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20The%20Fino%20Partners.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fthefinopartners.com%5C%2Fblogs%5C%2Ffixed-costs-vs-variable-costs-a-practical-guide-with-examples%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fthefinopartners.com%5C%2Fblogs%5C%2Ffixed-costs-vs-variable-costs-a-practical-guide-with-examples%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Fixed%20Costs%20vs%20Variable%20Costs%3A%20A%20Practical%20Guide%20with%20Examples%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22A%20fixed%20cost%20is%20any%20business%20expense%20that%20doesn%26%23039%3Bt%20change%20with%20the%20number%20of%20units%20your%20business%20produces%2C%20sales%20it%20makes%2C%20or%20revenue%20it%20generates.%20A%20fixed%20cost%20is%20a%20firm%20expense%20you%20can%20predict%20every%20single%20time.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fthefinopartners.com%5C%2Fblogs%5C%2Ffixed-costs-vs-variable-costs-a-practical-guide-with-examples%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T16%3A16%3A05Z%22%7D%7D%2C%7B%22key%22%3A%22PJT2KBBP%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22wtolson1%22%2C%22parsedDate%22%3A%222025-11-03%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3Bwtolson1.%20%282025%2C%20November%203%29.%20R%26amp%3BD%20Tax%20Credits%20and%20Deductions%20Explained.%20%26lt%3Bi%26gt%3BBloomberg%20Tax%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fpro.bloombergtax.com%5C%2Finsights%5C%2Ffederal-tax%5C%2Frd-tax-credit-and-deducting-rd-expenditures%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fpro.bloombergtax.com%5C%2Finsights%5C%2Ffederal-tax%5C%2Frd-tax-credit-and-deducting-rd-expenditures%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22R%26D%20Tax%20Credits%20and%20Deductions%20Explained%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22wtolson1%22%7D%5D%2C%22abstractNote%22%3A%22R%26amp%3BD%20tax%20credits%20can%20save%20your%20organization%20thousands%20of%20dollars%20per%20year.%20Learn%20more%20about%20these%20credits%20and%20deductions%2C%20if%20you%20qualify%2C%20and%20more.%22%2C%22blogTitle%22%3A%22Bloomberg%20Tax%22%2C%22date%22%3A%222025-11-03T13%3A03%3A57%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fpro.bloombergtax.com%5C%2Finsights%5C%2Ffederal-tax%5C%2Frd-tax-credit-and-deducting-rd-expenditures%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A47%3A39Z%22%7D%7D%2C%7B%22key%22%3A%22NDQW9KSQ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ohnstad%22%2C%22parsedDate%22%3A%222024-07-31%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BOhnstad%2C%20E.%20%282024%2C%20July%2031%29.%20Can%20your%20business%20qualify%20for%20the%20R%26amp%3BD%20tax%20credit%3F%20%26lt%3Bi%26gt%3BAbdo%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fabdosolutions.com%5C%2Fcan-your-business-qualify-for-the-rd-tax-credit%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fabdosolutions.com%5C%2Fcan-your-business-qualify-for-the-rd-tax-credit%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Can%20your%20business%20qualify%20for%20the%20R%26D%20tax%20credit%3F%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elly%22%2C%22lastName%22%3A%22Ohnstad%22%7D%5D%2C%22abstractNote%22%3A%22Unlock%20the%20potential%20of%20Research%20%26amp%3B%20Development%20Tax%20Credits%20for%20your%20industry.%20Explore%20how%20businesses%20in%20manufacturing%2C%20building%20%26amp%3B%20remodeling%2C%20software%2C%20engineering%2C%20and%20more%20can%20qualify%20for%20R%26amp%3BD%20tax%20credits.%22%2C%22blogTitle%22%3A%22Abdo%22%2C%22date%22%3A%222024-07-31T18%3A30%3A32%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fabdosolutions.com%5C%2Fcan-your-business-qualify-for-the-rd-tax-credit%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A47%3A35Z%22%7D%7D%2C%7B%22key%22%3A%2225FX7ZRB%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Gibson%22%2C%22parsedDate%22%3A%222023-03-03%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BGibson%2C%20T.%20%282023%2C%20March%203%29.%205%20Advantages%20of%20the%20Small%20Business%20R%26amp%3BD%20Tax%20Credit.%20%26lt%3Bi%26gt%3BOmega%20Accounting%20Solutions%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fomega-accounting.com%5C%2Fsmall-business-r-d-tax-credit%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fomega-accounting.com%5C%2Fsmall-business-r-d-tax-credit%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%225%20Advantages%20of%20the%20Small%20Business%20R%26D%20Tax%20Credit%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Timothy%22%2C%22lastName%22%3A%22Gibson%22%7D%5D%2C%22abstractNote%22%3A%22R%26amp%3BD%20tax%20credits%20are%20for%20companies%20of%20all%20sizes%2C%20and%20you%20don%5Cu2019t%20have%20to%20cure%20cancer%20to%20qualify.%20Learn%20why%20small%20businesses%20may%20be%20eligible.%22%2C%22blogTitle%22%3A%22Omega%20Accounting%20Solutions%22%2C%22date%22%3A%222023-03-03T00%3A00%3A00%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fomega-accounting.com%5C%2Fsmall-business-r-d-tax-credit%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A47%3A31Z%22%7D%7D%2C%7B%22key%22%3A%2275XH59FV%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Council%22%2C%22parsedDate%22%3A%222025-08-04%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BCouncil%2C%20S.%20B.%20E.%20%282025%2C%20August%204%29.%20%26%23x201C%3BOne%20Big%20Beautiful%20Bill%26%23x201D%3B%20Encourages%20Small%20Business%20Innovation%3A%20How%20Entrepreneurs%20Can%20Immediately%20Benefit%20from%20Restored%20R%26amp%3BD%20Expensing.%20%26lt%3Bi%26gt%3BSmall%20Business%20%26amp%3B%20Entrepreneurship%20Council%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fsbecouncil.org%5C%2F2025%5C%2F08%5C%2F04%5C%2Fone-big-beautiful-bill-encourages-small-business-innovation-how-entrepreneurs-can-immediately-benefit-from-restored-rd-expensing%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fsbecouncil.org%5C%2F2025%5C%2F08%5C%2F04%5C%2Fone-big-beautiful-bill-encourages-small-business-innovation-how-entrepreneurs-can-immediately-benefit-from-restored-rd-expensing%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22%5Cu201cOne%20Big%20Beautiful%20Bill%5Cu201d%20Encourages%20Small%20Business%20Innovation%3A%20How%20Entrepreneurs%20Can%20Immediately%20Benefit%20from%20Restored%20R%26D%20Expensing%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22S.%20B.%20E.%22%2C%22lastName%22%3A%22Council%22%7D%5D%2C%22abstractNote%22%3A%22By%20Barbara%20Weltman%20%5Cu2013%20You%20don%5Cu2019t%20have%20to%20be%20in%20the%20pharmaceutical%20industry%20or%20develop%20software%20to%20engage%20in%20research%20and%20development%20%28R%26amp%3BD%29%20activities%20that%20can%20produce%20new%20things%20for%20sale%20or%20create%20new%20processes%20for%20your%20internal%20business%20operations.%20If%20you%20spend%20money%20on%20R%26amp%3BD%2C%20you%20can%20recoup%20some%20of%20your%20costs%20through%20federal%20%5B%5Cu2026%5D%22%2C%22blogTitle%22%3A%22Small%20Business%20%26%20Entrepreneurship%20Council%22%2C%22date%22%3A%222025-08-04T19%3A56%3A09%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fsbecouncil.org%5C%2F2025%5C%2F08%5C%2F04%5C%2Fone-big-beautiful-bill-encourages-small-business-innovation-how-entrepreneurs-can-immediately-benefit-from-restored-rd-expensing%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A47%3A26Z%22%7D%7D%2C%7B%22key%22%3A%227SP6I8E5%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BWater%20Quality%20Testing%20Business%20Plan%20Report%202026%20%7C%20Setup%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.imarcgroup.com%5C%2Fwater-quality-testing-business-plan-project-report%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.imarcgroup.com%5C%2Fwater-quality-testing-business-plan-project-report%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Water%20Quality%20Testing%20Business%20Plan%20Report%202026%20%7C%20Setup%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22How%20to%20start%20water%20quality%20testing%20business%3F%20Explore%20buisness%20plan%2C%20industry%20trends%2C%20business%20setup%2C%20revenue%20model%2C%20investment%2C%20expenses%20%26amp%3B%20profitability.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.imarcgroup.com%5C%2Fwater-quality-testing-business-plan-project-report%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A47%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22ELSX67YZ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Brockovich%22%2C%22parsedDate%22%3A%222025-11-12%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BBrockovich%2C%20E.%20%282025%2C%20November%2012%29.%20%26lt%3Bi%26gt%3BWho%26%23x2019%3Bs%20In%20Charge%20Of%20The%20Water%20In%20Madison%20County%2C%20Indiana%3F%26lt%3B%5C%2Fi%26gt%3B%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.thebrockovichreport.com%5C%2Fp%5C%2Fwhos-in-charge-of-the-water-in-madison%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.thebrockovichreport.com%5C%2Fp%5C%2Fwhos-in-charge-of-the-water-in-madison%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Who%27s%20In%20Charge%20Of%20The%20Water%20In%20Madison%20County%2C%20Indiana%3F%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Erin%22%2C%22lastName%22%3A%22Brockovich%22%7D%5D%2C%22abstractNote%22%3A%22Local%20Officials%20Fumble%20As%20Residents%20Raise%20Concerns%20About%20The%20Look%20Of%20Their%20Water%22%2C%22date%22%3A%222025-11-12%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.thebrockovichreport.com%5C%2Fp%5C%2Fwhos-in-charge-of-the-water-in-madison%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A43%3A45Z%22%7D%7D%2C%7B%22key%22%3A%22BVVF7B89%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Amritha%20R%22%2C%22parsedDate%22%3A%222024-09-22%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BAmritha%20R.%20%282024%2C%20September%2022%29.%20%26lt%3Bi%26gt%3BTHE%20IMPACT%20OF%20VARIABLE%20AND%20FIXED%20COST%20ON%20BUSINESS%20DECISION-MAKING%20%7C%20LinkedIn%26lt%3B%5C%2Fi%26gt%3B%20%5BSocial%20Media%5D.%20Geojit%20Investments%20Limited.%20LinkedIn.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.linkedin.com%5C%2Fpulse%5C%2Fimpact-variable-fixed-cost-business-decision-making-amritha-r-3axbc%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.linkedin.com%5C%2Fpulse%5C%2Fimpact-variable-fixed-cost-business-decision-making-amritha-r-3axbc%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22THE%20IMPACT%20OF%20VARIABLE%20AND%20FIXED%20COST%20ON%20BUSINESS%20DECISION-MAKING%20%7C%20LinkedIn%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22Amritha%20R%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%222024-09-22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.linkedin.com%5C%2Fpulse%5C%2Fimpact-variable-fixed-cost-business-decision-making-amritha-r-3axbc%5C%2F%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A35%3A55Z%22%7D%7D%2C%7B%22key%22%3A%22ZD9SIRUP%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22365outsource_admin%22%2C%22parsedDate%22%3A%222025-10-24%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B365outsource_admin.%20%282025%2C%20October%2024%29.%20How%20Small%20Businesses%20Cut%20Costs%20with%20Outsourcing.%20%26lt%3Bi%26gt%3B365Outsource.Com%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.365outsource.com%5C%2Funcategorized%5C%2Fsmall-businesses-cut-costs-outsourcing%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.365outsource.com%5C%2Funcategorized%5C%2Fsmall-businesses-cut-costs-outsourcing%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22How%20Small%20Businesses%20Cut%20Costs%20with%20Outsourcing%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22365outsource_admin%22%7D%5D%2C%22abstractNote%22%3A%22Learn%20how%20small%20businesses%20can%20save%20costs%2C%20access%20expertise%2C%20and%20enhance%20efficiency%20through%20strategic%20outsourcing.%22%2C%22blogTitle%22%3A%22365Outsource.com%22%2C%22date%22%3A%222025-10-24T04%3A19%3A53%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.365outsource.com%5C%2Funcategorized%5C%2Fsmall-businesses-cut-costs-outsourcing%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A18%3A15Z%22%7D%7D%2C%7B%22key%22%3A%22J2K44NWR%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Sullivan%22%2C%22parsedDate%22%3A%222023-09-19%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BSullivan%2C%20P.%20%282023%2C%20September%2019%29.%20Why%20You%20Need%20a%20Third-Party%20Assessment%20for%20Cybersecurity.%20%26lt%3Bi%26gt%3BA-LIGN%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.a-lign.com%5C%2Farticles%5C%2Fwhy-you-need-a-third-party-assessment-for-cybersecurity%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.a-lign.com%5C%2Farticles%5C%2Fwhy-you-need-a-third-party-assessment-for-cybersecurity%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Why%20You%20Need%20a%20Third-Party%20Assessment%20for%20Cybersecurity%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Patrick%22%2C%22lastName%22%3A%22Sullivan%22%7D%5D%2C%22abstractNote%22%3A%22While%20a%20self-assessment%20can%20be%20the%20initial%20start%20to%20a%20compliance%20journey%2C%20a%20third-party%20assessment%20is%20not%20just%20preferable%2C%20but%20essential.%22%2C%22blogTitle%22%3A%22A-LIGN%22%2C%22date%22%3A%222023-09-19T12%3A56%3A28%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.a-lign.com%5C%2Farticles%5C%2Fwhy-you-need-a-third-party-assessment-for-cybersecurity%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A12%3A08Z%22%7D%7D%2C%7B%22key%22%3A%22RWBMED99%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BWhat%20Is%20Third-Party%20Risk%20Management%3F%26lt%3B%5C%2Fi%26gt%3B%20%28n.d.%29.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.venminder.com%5C%2Fblog%5C%2Fwhat-is-third-party-risk-management%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.venminder.com%5C%2Fblog%5C%2Fwhat-is-third-party-risk-management%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22What%20Is%20Third-Party%20Risk%20Management%3F%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22Third-party%20risk%20management%20helps%20identify%2C%20assess%2C%20manage%2C%20%26amp%3B%20monitor%20vendor%20risks%20posed%20to%20your%20organization.%20Learn%20what%20third-party%20risk%20management%20is.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.venminder.com%5C%2Fblog%5C%2Fwhat-is-third-party-risk-management%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A12%3A03Z%22%7D%7D%2C%7B%22key%22%3A%22CGX4UKLZ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BWater%20Quality%20Studies%20%26%23x2013%3B%20Johnson%20Engineering%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fjohnsonengineering.com%5C%2Fservices-projects%5C%2Fwater-quality-studies%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fjohnsonengineering.com%5C%2Fservices-projects%5C%2Fwater-quality-studies%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Water%20Quality%20Studies%20%5Cu2013%20Johnson%20Engineering%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22%22%2C%22blogTitle%22%3A%22%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fjohnsonengineering.com%5C%2Fservices-projects%5C%2Fwater-quality-studies%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A09%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22XJQSDMLI%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22parsedDate%22%3A%222025-06-26%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BWater%20Treatment%20Feasibility%20Studies%20-%20AQUASGROUP%26lt%3B%5C%2Fi%26gt%3B.%20%282025%2C%20June%2026%29.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Faquasgroup.com%5C%2Fservice%5C%2Fion-exchange-resin-regeneration-activated-carbon-reactivation-services%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Faquasgroup.com%5C%2Fservice%5C%2Fion-exchange-resin-regeneration-activated-carbon-reactivation-services%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22Water%20Treatment%20Feasibility%20Studies%20-%20AQUASGROUP%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22ISO%209001%3A2015%20certified%20ion%20exchange%20resin%20regeneration%20and%20GAC%20reactivation%20services%20for%20industrial%20process%20water%20in%20aerospace%2C%20medical%20device%2C%20electronics%2C%20and%20more.%22%2C%22date%22%3A%222025-06-26T14%3A40%3A38%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Faquasgroup.com%5C%2Fservice%5C%2Fion-exchange-resin-regeneration-activated-carbon-reactivation-services%5C%2F%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A09%3A02Z%22%7D%7D%2C%7B%22key%22%3A%22GTUH53EI%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BLife%20Science%20Content%20Marketing%20Services.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BSciencia%20Consulting%26lt%3B%5C%2Fi%26gt%3B.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fscienciaconsulting.com%5C%2Fcontent-marketing-for-life-sciences%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fscienciaconsulting.com%5C%2Fcontent-marketing-for-life-sciences%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22Life%20Science%20Content%20Marketing%20Services%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22Boost%20your%20brand%20with%20expert%20life%20science%20content%20marketing.%20Sciencia%20Consulting%20delivers%20compliant%2C%20engaging%20content%20to%20drive%20traffic%20and%20trust.%22%2C%22blogTitle%22%3A%22Sciencia%20Consulting%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fscienciaconsulting.com%5C%2Fcontent-marketing-for-life-sciences%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T15%3A08%3A55Z%22%7D%7D%2C%7B%22key%22%3A%22VYKM956X%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B%26lt%3Bi%26gt%3BNew%20Evidence%20That%20the%20R%26amp%3BD%20Tax%20Credit%20Promotes%20Research%2C%20Especially%20in%20Small%20Companies%20%7C%20ITIF%26lt%3B%5C%2Fi%26gt%3B.%20%28n.d.%29.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fitif.org%5C%2Fpublications%5C%2F2020%5C%2F05%5C%2F05%5C%2Fnew-evidence-rd-tax-credit-promotes-research-especially-small-companies%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fitif.org%5C%2Fpublications%5C%2F2020%5C%2F05%5C%2F05%5C%2Fnew-evidence-rd-tax-credit-promotes-research-especially-small-companies%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22New%20Evidence%20That%20the%20R%26D%20Tax%20Credit%20Promotes%20Research%2C%20Especially%20in%20Small%20Companies%20%7C%20ITIF%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fitif.org%5C%2Fpublications%5C%2F2020%5C%2F05%5C%2F05%5C%2Fnew-evidence-rd-tax-credit-promotes-research-especially-small-companies%5C%2F%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T14%3A44%3A54Z%22%7D%7D%2C%7B%22key%22%3A%22DCTFA8QU%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22parsedDate%22%3A%222016-03-08%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BThe%20Type%20of%20R%26amp%3BD%20That%20Small%20Businesses%20Can%20Really%20Use%20-%20NicheLabs%2C%20LLC.%20%282016%2C%20March%208%29.%20%26lt%3Bi%26gt%3BNicheLabs%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.nichelabs.com%5C%2Fgeneral%5C%2Fthe-type-of-rd-that-small-businesses-can-really-use%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.nichelabs.com%5C%2Fgeneral%5C%2Fthe-type-of-rd-that-small-businesses-can-really-use%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22The%20Type%20of%20R%26D%20That%20Small%20Businesses%20Can%20Really%20Use%20-%20NicheLabs%2C%20LLC%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22If%20you%20are%20looking%20to%20grow%20your%20business%20to%20find%20more%20clients%2C%20customers%20or%20patients%20without%20spending%20a%20fortune%2C%20then%20the%20tactic%20that%20my%20friend%20Steve%20Clements%22%2C%22blogTitle%22%3A%22NicheLabs%22%2C%22date%22%3A%222016-03-08T10%3A00%3A58-04%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.nichelabs.com%5C%2Fgeneral%5C%2Fthe-type-of-rd-that-small-businesses-can-really-use%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T14%3A44%3A43Z%22%7D%7D%2C%7B%22key%22%3A%22PUB8TJ97%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22parsedDate%22%3A%222016-03-08%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3BThe%20Type%20of%20R%26amp%3BD%20That%20Small%20Businesses%20Can%20Really%20Use%20-%20NicheLabs%2C%20LLC.%20%282016%2C%20March%208%29.%20%26lt%3Bi%26gt%3BNicheLabs%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.nichelabs.com%5C%2Fgeneral%5C%2Fthe-type-of-rd-that-small-businesses-can-really-use%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.nichelabs.com%5C%2Fgeneral%5C%2Fthe-type-of-rd-that-small-businesses-can-really-use%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22The%20Type%20of%20R%26D%20That%20Small%20Businesses%20Can%20Really%20Use%20-%20NicheLabs%2C%20LLC%22%2C%22creators%22%3A%5B%5D%2C%22abstractNote%22%3A%22If%20you%20are%20looking%20to%20grow%20your%20business%20to%20find%20more%20clients%2C%20customers%20or%20patients%20without%20spending%20a%20fortune%2C%20then%20the%20tactic%20that%20my%20friend%20Steve%20Clements%22%2C%22blogTitle%22%3A%22NicheLabs%22%2C%22date%22%3A%222016-03-08T10%3A00%3A58-04%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.nichelabs.com%5C%2Fgeneral%5C%2Fthe-type-of-rd-that-small-businesses-can-really-use%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T14%3A44%3A34Z%22%7D%7D%2C%7B%22key%22%3A%22DKN7V5GZ%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22editor%20et%20al.%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3Beditor%2C%20P.%20R.%20F.%20B.%20P.%20R.%20is%20a%20copy%2C%20Economics%2C%20F.-C.%20with%20E.%20in%2C%20Finance%2C%20P.%2C%20%26amp%3B%20policies%2C%20%20over%20twenty%20years%20of%20experience%20in%20the%20classroom%20L.%20about%20our%20editorial.%20%28n.d.%29.%20%26lt%3Bi%26gt%3BHow%20Outsourcing%20Reduces%20Business%20Costs%3A%20Strategies%20and%20Examples%26lt%3B%5C%2Fi%26gt%3B.%20Investopedia.%20Retrieved%20February%205%2C%202026%2C%20from%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.investopedia.com%5C%2Fterms%5C%2Fo%5C%2Foutsourcing.asp%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.investopedia.com%5C%2Fterms%5C%2Fo%5C%2Foutsourcing.asp%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22webpage%22%2C%22title%22%3A%22How%20Outsourcing%20Reduces%20Business%20Costs%3A%20Strategies%20and%20Examples%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pete%20Rathburn%20Full%20Bio%20Pete%20Rathburn%20is%20a%20copy%22%2C%22lastName%22%3A%22editor%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fact-Checker%20with%20Expertise%20in%22%2C%22lastName%22%3A%22Economics%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Personal%22%2C%22lastName%22%3A%22Finance%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22over%20twenty%20years%20of%20experience%20in%20the%20classroom%20Learn%20about%20our%20editorial%22%2C%22lastName%22%3A%22policies%22%7D%5D%2C%22abstractNote%22%3A%22Discover%20how%20outsourcing%20can%20lower%20business%20costs%20and%20enhance%20efficiency.%20Learn%20strategies%20with%20practical%20examples%2C%20benefits%2C%20and%20potential%20downsides%20of%20outsourcing.%22%2C%22date%22%3A%22%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.investopedia.com%5C%2Fterms%5C%2Fo%5C%2Foutsourcing.asp%22%2C%22language%22%3A%22en%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T14%3A41%3A39Z%22%7D%7D%2C%7B%22key%22%3A%22PWPNYP7W%22%2C%22library%22%3A%7B%22id%22%3A21495%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22365outsource_admin%22%2C%22parsedDate%22%3A%222025-10-24%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%26lt%3Bdiv%20class%3D%26quot%3Bcsl-bib-body%26quot%3B%20style%3D%26quot%3Bline-height%3A%202%3B%20padding-left%3A%201em%3B%20text-indent%3A-1em%3B%26quot%3B%26gt%3B%5Cn%20%20%26lt%3Bdiv%20class%3D%26quot%3Bcsl-entry%26quot%3B%26gt%3B365outsource_admin.%20%282025%2C%20October%2024%29.%20How%20Small%20Businesses%20Cut%20Costs%20with%20Outsourcing.%20%26lt%3Bi%26gt%3B365Outsource.Com%26lt%3B%5C%2Fi%26gt%3B.%20%26lt%3Ba%20class%3D%26%23039%3Bzp-ItemURL%26%23039%3B%20href%3D%26%23039%3Bhttps%3A%5C%2F%5C%2Fwww.365outsource.com%5C%2Funcategorized%5C%2Fsmall-businesses-cut-costs-outsourcing%5C%2F%26%23039%3B%26gt%3Bhttps%3A%5C%2F%5C%2Fwww.365outsource.com%5C%2Funcategorized%5C%2Fsmall-businesses-cut-costs-outsourcing%5C%2F%26lt%3B%5C%2Fa%26gt%3B%26lt%3B%5C%2Fdiv%26gt%3B%5Cn%26lt%3B%5C%2Fdiv%26gt%3B%22%2C%22data%22%3A%7B%22itemType%22%3A%22blogPost%22%2C%22title%22%3A%22How%20Small%20Businesses%20Cut%20Costs%20with%20Outsourcing%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22name%22%3A%22365outsource_admin%22%7D%5D%2C%22abstractNote%22%3A%22Learn%20how%20small%20businesses%20can%20save%20costs%2C%20access%20expertise%2C%20and%20enhance%20efficiency%20through%20strategic%20outsourcing.%22%2C%22blogTitle%22%3A%22365Outsource.com%22%2C%22date%22%3A%222025-10-24T04%3A19%3A53%2B00%3A00%22%2C%22DOI%22%3A%22%22%2C%22citationKey%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.365outsource.com%5C%2Funcategorized%5C%2Fsmall-businesses-cut-costs-outsourcing%5C%2F%22%2C%22ISSN%22%3A%22%22%2C%22language%22%3A%22en-US%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222026-02-05T14%3A41%3A35Z%22%7D%7D%5D%7D
National Research Council (U.S.). (2012). A framework for K-12 science education: practices, crosscutting concepts, and core ideas . The National Academies Press.
Freeman, S., Eddy, S. L., McDonough, M., Smith, M. K., Okoroafor, N., Jordt, H., & Wenderoth, M. P. (2014). Active learning increases student performance in science, engineering, and mathematics.
Proceedings of the National Academy of Sciences ,
111 (23), 8410–8415.
https://doi.org/10.1073/pnas.1319030111
Jimenez, B. A., Croft, G., Twine, J., & Gorey, J. (2021). Development of Engineering Habits of Mind for Students With Intellectual Disability.
The Journal of Special Education ,
55 (3), 174–185.
https://doi.org/10.1177/00224669211009960
Centre for Real-World Learning. (2014, May). Thinking like an engineer
Implications for the
education system . Royal Academy of Engineering.
Cochrane, P. (2021, November 10).
Private Wells & Testing Drinking Water [Video recording].
https://youtu.be/9mtMqsTiNag?si=o2ax8328ItI-2GNu
NRCS. (n.d.). Soil Testing for Small Farms and Gardens [Technical Notes]. USDA.
Paul Cochrane. (2021, November 10).
Private Wells & Testing Drinking Water [Video recording].
https://www.youtube.com/watch?v=9mtMqsTiNag
US EPA, O. (2015, May 6).
Protect Your Home’s Water [Overviews and Factsheets].
https://www.epa.gov/privatewells/protect-your-homes-water
Douglas, R. (2021, May 11).
Soil Test Information | N.C. Cooperative Extension . Haywood County Center.
https://haywood.ces.ncsu.edu/soil-test-information/
Jones, M. (2014, August 20).
Soil Testing for Lawns and Gardens | N.C. Cooperative Extension . Chatham County Center.
https://chatham.ces.ncsu.edu/soil-testing-for-lawns-and-gardens/
A Gardener’s Guide to Soil Testing | NC State Extension Publications . (n.d.). Retrieved March 2, 2026, from
https://content.ces.ncsu.edu/a-gardeners-guide-to-soil-testing
Soil Testing . (n.d.). Purdue University - Extension. Retrieved March 2, 2026, from
https://extension.purdue.edu/county/marion/anr/soil-testing.html
Proton Mail . (2026, March 8).
https://mail.proton.me/
anubha. (2024, November 11). Lab Work Made Easy: Choosing the Right Chemistry Glassware.
Scientific Lab Equipment Manufacturer and Supplier .
https://www.shivsons.com/lab-work-made-easy-choosing-the-right-chemistry-glassware/
The4. (2026, January 29).
A list of glassware equipment for high school chemistry lab. - Science . Science Equip.
https://www.scienceequip.com.au/blogs/news/a-list-of-glassware-equipment-for-high-school-chemistry-lab
says, G. johnson. (n.d.).
Beakers vs. Flasks: The Pros and Cons of Frequently Used Lab Glassware . Lab Pro Inc. Retrieved February 26, 2026, from
https://labproinc.com/blogs/laboratory-equipment/beakers-vs-flasks-the-pros-and-cons-of-frequently-used-lab-glassware
Understanding Laboratory Glassware Classifications . (n.d.). Globe Scientific. Retrieved February 26, 2026, from
https://globescientific.com/blogs/blog/understanding-laboratory-glassware-classifications
Laboratory Beakers Guide – Types & Uses Explained . (n.d.). ChemScience Inc. Retrieved February 26, 2026, from
https://www.chemscience.com/blog/www.chemscience.com
Lam, K., Armstrong, B., & Margelefsky, E. L. (2024). Correction to “Gravimetric and Thermal-Imaging Characterization of Water-Free Reflux Condensers for Laboratory Applications.”
ACS Omega ,
9 (20), 22508.
https://doi.org/10.1021/acsomega.4c03508
Lam, K., Armstrong, B., & Margelefsky, E. L. (2024). Gravimetric and Thermal-Imaging Characterization of Water-Free Reflux Condensers for Laboratory Applications.
ACS Omega ,
9 (14), 16563–16571.
https://doi.org/10.1021/acsomega.4c00411
Fordham, C. (2024, February 7). Condenser Chemistry: What’s the Difference Between Distillation & Reflux?
Asynt .
https://www.asynt.com/blog/condenser-chemistry-difference-between-distillation-reflux/
Fordham, C. (2024, February 7). Condenser Chemistry: What’s the Difference Between Distillation & Reflux?
Asynt .
https://www.asynt.com/blog/condenser-chemistry-difference-between-distillation-reflux/
admin. (2025, October 28). What Is Reflux Distillation? A Complete Guide.
Borosil Scientific .
https://www.borosilscientific.com/what-is-reflux-distillation-complete-guide/
Reflux and distillation. (n.d.).
Reflux and distillation – practical videos | 16–18 students . RSC Education. Retrieved February 18, 2026, from
https://edu.rsc.org/practical/reflux-and-distillation-practical-videos-16-18-students/4012294.article
Water flow in condenser - ECHEMI.com . (n.d.). ECHEMI. Retrieved February 18, 2026, from
https://www.echemi.com/community/water-flow-in-condenser_mjart2205112471_431.html
Abrahams, I., & Millar, R. (2008). Does Practical Work Really Work? A study of the effectiveness of practical work as a teaching and learning method in school science.
International Journal of Science Education ,
30 (14), 1945–1969.
https://doi.org/10.1080/09500690701749305
Vogel, A. I. (1977). A text-book of practical organic chemistry: incl. qualitative organic analysis (3. ed.; new impr). Longmans.
Indiana, W. Q. in. (2025, October 22).
Community Drinking Water Information - Madison County . Water Quality in Indiana.
https://www.in.gov/idem/cleanwater/drinking-water/community-drinking-water-info-madison-co/
Schneider, K. (n.d.).
These Indiana residents have brown water and want their town to tell them why . The Indianapolis Star. Retrieved February 5, 2026, from
https://www.indystar.com/story/news/environment/2025/10/06/indiana-brown-water-quality-safe-drinking/86481832007/
Fixed Costs vs Variable Costs: A Practical Guide with Examples . (n.d.). The Fino Partners. Retrieved February 5, 2026, from
https://thefinopartners.com/blogs/fixed-costs-vs-variable-costs-a-practical-guide-with-examples
Ohnstad, E. (2024, July 31). Can your business qualify for the R&D tax credit?
Abdo .
https://abdosolutions.com/can-your-business-qualify-for-the-rd-tax-credit/
Gibson, T. (2023, March 3). 5 Advantages of the Small Business R&D Tax Credit.
Omega Accounting Solutions .
https://omega-accounting.com/small-business-r-d-tax-credit/
Council, S. B. E. (2025, August 4). “One Big Beautiful Bill” Encourages Small Business Innovation: How Entrepreneurs Can Immediately Benefit from Restored R&D Expensing.
Small Business & Entrepreneurship Council .
https://sbecouncil.org/2025/08/04/one-big-beautiful-bill-encourages-small-business-innovation-how-entrepreneurs-can-immediately-benefit-from-restored-rd-expensing/
Water Quality Testing Business Plan Report 2026 | Setup . (n.d.). Retrieved February 5, 2026, from
https://www.imarcgroup.com/water-quality-testing-business-plan-project-report
Brockovich, E. (2025, November 12).
Who’s In Charge Of The Water In Madison County, Indiana? https://www.thebrockovichreport.com/p/whos-in-charge-of-the-water-in-madison
Amritha R. (2024, September 22).
THE IMPACT OF VARIABLE AND FIXED COST ON BUSINESS DECISION-MAKING | LinkedIn [Social Media]. Geojit Investments Limited. LinkedIn.
https://www.linkedin.com/pulse/impact-variable-fixed-cost-business-decision-making-amritha-r-3axbc/
365outsource_admin. (2025, October 24). How Small Businesses Cut Costs with Outsourcing.
365Outsource.Com .
https://www.365outsource.com/uncategorized/small-businesses-cut-costs-outsourcing/
Sullivan, P. (2023, September 19). Why You Need a Third-Party Assessment for Cybersecurity.
A-LIGN .
https://www.a-lign.com/articles/why-you-need-a-third-party-assessment-for-cybersecurity
What Is Third-Party Risk Management? (n.d.). Retrieved February 5, 2026, from
https://www.venminder.com/blog/what-is-third-party-risk-management
Water Quality Studies – Johnson Engineering . (n.d.). Retrieved February 5, 2026, from
https://johnsonengineering.com/services-projects/water-quality-studies/
Life Science Content Marketing Services. (n.d.).
Sciencia Consulting . Retrieved February 5, 2026, from
https://scienciaconsulting.com/content-marketing-for-life-sciences/
New Evidence That the R&D Tax Credit Promotes Research, Especially in Small Companies | ITIF . (n.d.). Retrieved February 5, 2026, from
https://itif.org/publications/2020/05/05/new-evidence-rd-tax-credit-promotes-research-especially-small-companies/
The Type of R&D That Small Businesses Can Really Use - NicheLabs, LLC. (2016, March 8).
NicheLabs .
https://www.nichelabs.com/general/the-type-of-rd-that-small-businesses-can-really-use/
The Type of R&D That Small Businesses Can Really Use - NicheLabs, LLC. (2016, March 8).
NicheLabs .
https://www.nichelabs.com/general/the-type-of-rd-that-small-businesses-can-really-use/
editor, P. R. F. B. P. R. is a copy, Economics, F.-C. with E. in, Finance, P., & policies, over twenty years of experience in the classroom L. about our editorial. (n.d.).
How Outsourcing Reduces Business Costs: Strategies and Examples . Investopedia. Retrieved February 5, 2026, from
https://www.investopedia.com/terms/o/outsourcing.asp
365outsource_admin. (2025, October 24). How Small Businesses Cut Costs with Outsourcing.
365Outsource.Com .
https://www.365outsource.com/uncategorized/small-businesses-cut-costs-outsourcing/